抽象
我们进行了一项随机、双盲、II期、剂量增加试验,以评估单独使用酪氨酸激酶抑制剂nintedanib,并将其添加到正在进行的吡非尼酮治疗中的日本特发性肺纤维化患者的安全性、耐受性和药代动力学。
50名日本患者随机接受nintedanib或安慰剂治疗(nintedanib每天两次50毫克或每天两次100毫克,持续14天,或每天两次150毫克,持续28天)。纳入时接受吡非尼酮治疗的患者被分成nintedanib剂量组和安慰剂组。
17例接受尼替丹单药治疗的患者中有9例出现不良反应,21例接受尼替丹单加吡非尼酮治疗的患者中有10例出现不良反应。所有不良事件的强度均为轻度或中度。胃肠道疾病是最常见的不良事件。与吡非尼酮相比,nintedanib及其代谢物稳态时的最大血药浓度和曲线下面积趋于较低。尼坦对吡非尼酮的药动学无影响。
总之,需要进一步研究时,患者的特发性肺纤维化添加到吡非尼酮评估尼达尼布的安全性和耐受性。有向尼达尼布的低曝光趋势时,它被添加到吡非尼酮。
抽象
Nintedanib在日本IPF患者中具有可接受的安全性和耐受性http://ow.ly/DNG4k
介绍
特发性肺纤维化(Idiopathic pulmonary fibrosis, IPF)是一种以肺泡和肺间质纤维化为特征的慢性进行性肺间质性疾病[1,2]。Estimates of the worldwide prevalence of IPF range from two to 43 cases per 100 000 people [2]。IPF具有不良预后[2]并且存在临床需要新的疗法在IPF患者改善预后。
据推测,IPF的发病机制与上皮损伤引起的伤口愈合异常有关[3]。新治疗方法的发展主要集中在参与这种反应的信号通路。尼达尼布(以前称为BIBF 1120;勃林格殷格翰制药有限公司&Co. KG的,殷格翰,德国)是酪氨酸激酶的强效细胞内抑制剂已被用于治疗IPF的和一些癌症类型的显影。尼达尼布块的血小板衍生的生长因子的激酶活性,血管内皮生长因子和成纤维细胞生长因子受体,所有这些已显示出参与了纤维化的发展[4,五]。BIBF-1120改善肺纤维化II期(明天)试验结果提示,nintedanib 150 mg每日2次,可减少IPF患者肺功能下降,急性加重期减少,并维持与健康相关的生活质量[6]。最近,两个重复相III INPULSIS试验的结果表明,在尼达尼布IPF患者减少疾病进展通过显著减少用力肺活量(FVC)[的下降速度7]。在与尼达尼布治疗的患者中,最常见的不良反应是胃肠道功能紊乱,其中占多数研究药物的停药由于不良eventss [7]。
吡非尼酮(盐野义制药(日本大阪)和InterMune公司公司(布里斯班,CA,USA))在2008年(盐野义制药公司)被批准在日本IPF治疗和用于治疗成年患者有轻度至moderate IPF in the European Union in 2011. In the phase III CAPACITY (Clinical Studies Assessing Pirfenidone in IPF: Research of Efficacy and Safety Outcomes) 2 trial, 72 weeks' treatment with pirfenidone 2403 mg·day-1显著降低了IPF患者的FVC预测百分比的下降与安慰剂,但在并发CAPACITY 1试验中,从安慰剂显著差异,未观察到[8]。近日,52周,吡非尼酮的第三阶段评估来确保疗效和安全性特发性肺纤维化(ASCEND)试验表明吡非尼酮的好处与从基线到预测FVC百分比变化的安慰剂[9]。一世n a phase III study in Japanese patients with IPF, 52 weeks' treatment with pirfenidone 180 mg·day-1与安慰剂组相比,显著降低了肺活量的下降,匹非尼酮180 mg·d调整后的平均肺活量变化为-0.09 L-1与安慰剂组的-0.16 L比较[10]。在III期研究中,光敏性是日本患者最常见的不良事件,51.4%的患者使用吡非尼酮180 mg·d-1与22.4%服用安慰剂的病人[10]。然而,在国际能力和ASCEND试验中,不良事件与吡非尼酮治疗的患者报告的最常见的是恶心[8,9]。最近在基于与2059人暴露年累计曝光的数据集吡非尼酮治疗的患者报告的不良事件的评论报道,恶心和呕吐了40%和18%的报道,分别用吡非尼酮治疗的患者[11]。
有在IPF患者吡非尼酮和尼达尼布的不良事件谱在肝脏酶的胃肠活动,并增加条款[一些重叠6-9]。然而,吡非尼酮和nintedanib有不同的代谢特征;吡非尼酮被多种细胞色素P450酶代谢并以排泄为主通过尿作为主要代谢物5-羧基吡非尼酮[12,而nintedanib的代谢主要特征是产生BIBF 1202 ZW的酯裂解。BIBF 1202 ZW随后在肠道中被多种尿苷二磷酸葡萄糖醛酸转移酶(UGTs)和在肝脏中被UGT1A1所取代,形成BIBF 1202葡萄糖醛酸苷。nintedanib的两种代谢物都被排出体外通过胆道系统进入粪便[13]。
II期研究,以确定安全性,耐受性和添加到正在进行的吡非尼酮治疗的尼达尼布及其代谢产物,单独和药代动力学的时候,在日本IPF患者。吡非尼酮(单独和当尼达尼布物共同给药)的药动学也进行了研究。
材料和方法
研究对象
耐心eligible for inclusion were aged ≥40 years, with IPF diagnosed according to the American Thoracic Society/European Respiratory Society consensus statement [1]和第四版《日本特发性间质性肺炎诊断指南》[14<5年前进行筛查,并在随机化后12个月内进行高分辨率计算机断层扫描,这与诊断的IPF相一致。纳入标准包括植被覆盖度≥预测值的50%(根据日本预测正态方程)[15]和单次呼吸一氧化碳弥散量(dLCO),占预测值的30-79%。排除标准:肝酶升高(>为正常范围上限的1.5倍);有关气道阻塞;要求≥15小时·天-1补充氧气;treatment with oral corticosteroids at unstable doses or in excess of the equivalent of prednisone 15 mg·day-1,或与酮康唑或阿他扎那韦合用;期待进行肺移植或有迅速恶化的疾病;或预期寿命少于3个月。接受稳定剂量吡非尼酮≥3个月的患者有入选资格,并根据预先指定的分层方案(在下一节中描述)入组。
该试验符合赫尔辛基宣言(1996年10月)进行,并按照上协调良好的临床实践(GCP)的指导方针和日本GCP法规的国际会议(卫生和福利条例数28部; 3月27日,1997年)。所有患者都提供了进入研究前签署知情同意书。患者的安全是由一个独立的数据监察委员会(IDMC)监控。该试验在注册www.clinicaltrials.gov标识号NCT01136174。
学习规划
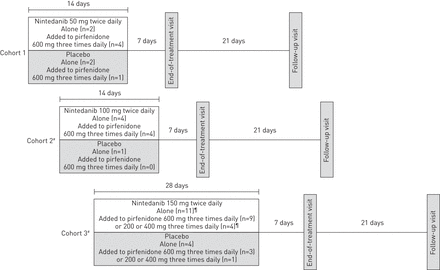
该研究是一项随机,双盲(相对于尼达尼布),安慰剂对照(剂量组内),多中心,剂量递增试验。一世t consisted of a screening visit occurring 5–21 days before the start of treatment, a 14-day (cohorts 1 and 2) or 28-day (cohort 3) treatment period, an end-of-treatment visit 7 days after the last dose (or on premature treatment discontinuation) and a follow-up visit 3 weeks after the end-of-treatment visit (图1)。治疗期间包括4次(队列1和2)或6次(队列3)在nintedanib开始治疗后进行药代动力学抽样和安全性评估。
研究设计和患者分布。#:病人的安全是由一个独立的数据监察委员会前患者被转移到这些同伙审查;¶:两名患者在研究完成前退出。
采用逐步方法将患者随机分为三组,在转入高剂量组之前进行IDMC安全性评估。队列1的患者随机接受nintedanib 50毫克每日两次或安慰剂治疗14天;2组患者随机接受nintedanib 100 mg每日两次或安慰剂治疗14天;3组患者随机加入nintedanib 150 mg,每日2次,或安慰剂,共28天(图3)。1)。耐心who had been receiving a steady dose of pirfenidone 600 mg three times daily for ≥3 months prior to inclusion were stratified to each cohort, and were included in the safety and pharmacokinetic analyses. Patients on a steady dose of pirfenidone 200 or 400 mg three times daily for ≥3 months prior to inclusion were allowed to participate in cohort 3 only and were included in safety analyses, but not in the pharmacokinetic analysis. It was planned that approximately half of every nintedanib and placebo group would be taking pirfenidone, except the placebo group in cohort 3, in which it was planned that two-thirds of the patients would be receiving pirfenidone. Pirfenidone was given as prescribed, after breakfast, lunch and dinner.
安全分析
所有治疗的患者被纳入安全性分析。Ťhe safety end-points assessed were the incidence and intensity (mild, moderate or severe) of adverse events, withdrawal due to adverse events, routine laboratory tests, lung function measurements (FVC, forced expiratory volume in 1 s (FEV1) anddLCO),血压和脉搏率。Analyses of adverse events focused on treatment-emergent events, defined as those occurring up to 28 days after the completion of study medication, or starting before the first drug intake and deteriorating during treatment. Laboratory tests, lung function and vital signs were measured at screening, baseline, on days 2, 7 and 14 (plus days 21 and 28 for cohort 3) of treatment, and at the end-of-treatment visit. Safety results for patients receiving placebo with or without pirfenidone were grouped across all cohorts.
药代动力学分析
为了定量研究nintedanib的血浆浓度、代谢物(BIBF 1202 ZW和BIBF 1202 glucuronide)和吡非尼酮(pirfenidone),我们在研究来访期间(给药前和给药后30分钟至72小时的稳定状态)收集了约4 mL静脉血。早餐和午餐剂量(给药前和给药后30分钟至12小时的时间点)前后均取样本进行吡非尼酮的药代动力学评估。nintedanib及其代谢物和吡非尼酮的血浆浓度采用经验证的液相色谱-串联质谱法测定(Nuvisan Pharma Services GmbH & Co. KG, neuu - ulm,德国)。
为尼达尼布和吡非尼酮计算的药代动力学参数包括:在上形成均匀给药间隔τ的稳态血浆中的分析物的浓度 - 时间曲线下的面积(AUCτ,SS)和在从t1至t2的时间间隔(AUCt1-t2中,SS);在稳定状态下在均匀的给药间隔τ(的C max,β)在血浆中的分析物的最大浓度;从在稳定状态下在均匀的给药间隔τ(Tmax时,SS)给药于分析物的最大浓度在血浆中的时间;在稳定状态下(T1 / 2,β)在血浆中分析物的末端半衰期;在稳定状态下血管外施用后,分析物在血浆中的表观清除率(CL / F,SS)(尼达尼布只);终端相λZ期间表观分布容积以下在稳态下(1/2 / F,SS)(尼达尼布只)施用血管外;基于AUCt1-t2中,SS和C max,SS(尼达尼布只)累积比率;和C max,ss和AUCt1-t2时,测试处理的SS到的C max,ss和AUCt1-T2中,参考治疗的SS(吡非尼酮只)的比率。药代动力学进行了评估尼达尼布及其代谢物,当它被单独给予,并且当它被添加到正在进行的吡非尼酮治疗(平行组比较),和有和没有尼达尼布共同给药(个体内比较)吡非尼酮。
结果
耐心
共有66名患者接受了8个地点的筛查,其中50人是随机的:6名患者随机接受nintedanib 50毫克每日两次,8名患者随机接受nintedanib 100毫克每日两次,24名患者随机接受nintedanib 150毫克每日两次,12名患者随机接受安慰剂(图1)。At screening, 26 patients were being treated with pirfenidone, five of whom entered cohort 1 (four randomised to nintedanib 50 mg twice daily and one to placebo), four entered cohort 2 (all randomised to nintedanib 100 mg twice daily) and 17 entered cohort 3 (13 randomised to nintedanib 150 mg twice daily and four to placebo) (图1)。在这些随机选择的患者中,46名(92.0%)患者完成了研究;每天两次150毫克组的4名患者过早地停药。各组间基线特征无显著差异(表1)。平均年龄为65.2岁,其中男性占70.0%。10例患者在基线接受了全身皮质激素治疗,4例接受了乙酰半胱氨酸治疗。
安全的结果
对所有50例随机患者进行安全性评估。共有23名(46.0%)患者经历了至少一次不良事件:4名(50.0%)患者接受nintedanib 100 mg每日两次,15名(62.5%)患者接受nintedanib 150 mg每日两次,4名(33.3%)患者接受安慰剂(表2)。不良事件是轻度的(87%)或在强度中等(13%)。Ťhe most common adverse events reported with nintedanib 150 mg twice daily were vomiting (five patients, all receiving nintedanib in addition to ongoing pirfenidone therapy) and nausea (five patients, four receiving nintedanib in addition to ongoing pirfenidone therapy). One serious adverse event (malignant hepatic neoplasm) was reported in the nintedanib 150 mg twice daily group during the follow-up period; this was not considered to be related to the study drug (previous computed tomography scans showed that the tumour was possibly present before study enrolment). Drug-related adverse events were reported by the investigator for 13 (26.0%) patients, most of which occurred in patients receiving nintedanib 150 mg twice daily (10 patients, seven receiving nintedanib in addition to ongoing pirfenidone therapy). The adverse events that led to discontinuation of study medication were increases in transaminase levels in three patients (all with nintedanib 150 mg twice daily, one with co-administration of pirfenidone 200 mg three times daily) and vomiting in one patient receiving nintedanib 150 mg twice daily and pirfenidone 200 mg three times daily. These adverse events resolved after treatment discontinuation. Photosensitivity was reported in one patient receiving pirfenidone alone and rash in one patient receiving pirfenidone with nintedanib 150 mg twice daily. FVC, FEV1 anddLCO在所有组中保持稳定。没有血压或脉搏率的一致变化,观察到的变化不认为与研究者的研究药物有关。
药代动力学结果
多次给药后,nintedanib的血浆浓度在给药后2-4小时达到最大值,在第7天达到稳定状态。多次给药后nintedanib的平均血浆浓度(第14天为队列1和2,第28天为队列3)如图所示图2。For the 150 mg twice daily dose, mean plasma concentrations of nintedanib were lower when nintedanib was added to ongoing pirfenidone therapy, but high variability was observed.
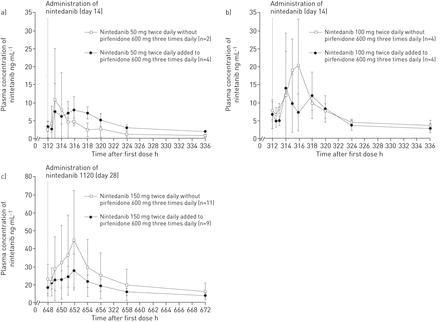
算术平均值±sd尼达尼布的血浆浓度 - 时间曲线在a)之后,每天两次(第14天)的尼达尼布50毫克多次给药,B)100 mg,每天两次(第14天)和c)150 mg,每天两次(单独给予,并加入到正在进行的第28天)吡非尼酮治疗(每日600毫克三次)。
经过28天的治疗nintedanib 150毫克每日两次,几何方法(Cmax gMean)、ss和AUCτ党卫军nintedanib低41% (23.5与39。7 ng·mL1)和32% (149与218 h·ng·mL1),当尼替丹加入正在进行的吡非尼酮治疗时,但个体值的分布重叠(图3)。对于gMean T1 / 2,ss和中位T max,当尼达尼布被单独给予时除了正在进行的吡非尼酮治疗给出为SS是相似的值(27.5与28.4小时和3.9小时与3。9 h, respectively) (表3)。中号ean CL/F,ss was 11 500 mL·min1in the absence of pirfenidone and 16 800 mL·min1当nintedanib加入吡非尼酮后,平均Vz/F,在没有吡非尼酮的情况下,ss为27300 L,加入正在进行的吡非尼酮(表3)。意思是RA,AUC他一直当添加到正在进行的吡非尼酮和平均值R是在不存在吡非尼酮的1.5和1.7A、C马克斯在没有吡非尼酮的情况下为1.1,在正在进行的吡非尼酮治疗中为1.3(表3)。吡非尼酮治疗对nintedanib的累积比率(RA、C马克斯和RA,AUC他一直) after multiple doses of nintedanib 150 mg twice daily (表3)。Ťhe effect of adding nintedanib to ongoing pirfenidone therapy on the pharmacokinetic parameters of nintedanib was similar for the nintedanib 100 mg twice daily dose as for the 150 mg twice daily dose (表3)。中号easurements of pharmacokinetic parameters were not obtained from enough patients receiving nintedanib 50 mg twice daily to adequately evaluate whether ongoing pirfenidone therapy had any effect.
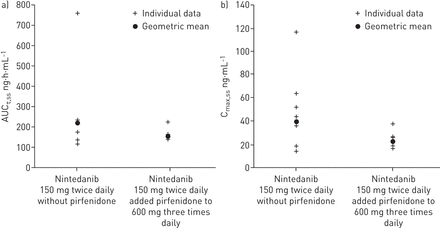
个人和几何平均曲线下的面积的分析物在稳态等离子体在一个统一的剂量间隔τ(AUCτ,ss)和最大浓度的分析物在稳态等离子体在一个统一的剂量间隔τ(Cmax, ss)值nintedanib 150毫克每日两次单独和添加到正在进行的pirfenidone治疗(每日三次,每次600毫克)。
单剂量nintedanib后,给药后nintedanib代谢物BIBF 1202 ZW和BIBF 1202 glucuronide的最大血浆浓度分别达到3-4 h和0.8-4.0 h, gMean t1/2,ss值分别约为23和46-56 h。在吡非尼酮治疗中加入nintedanib时,tmax、ss分别为BIBF 1202 ZW和BIBF 1202 glucuronide的3-5和0.7-8.0 h,而t1/2、ss为BIBF 1202 ZW的27-26 h和BIBF 1202 glucuronide的38-43 h。对于nintedanib 150毫克每日两次剂量,gMean Cmax,ss降低51% (15.4与33.2 ng·毫升-1)为BIBF 1202 ZW,低16% (107与128 ng·mL-1)在吡非尼酮治疗的基础上,使用nintedanib治疗BIBF 1202葡萄糖苷。AUCτ,党卫军(118低50%与237 h·ng·mL-1)为BIBF 1202 ZW和低20%(1100与1380 h·ng·mL-1)在吡非尼酮治疗的基础上,使用nintedanib治疗BIBF 1202葡萄糖苷。吡非尼酮治疗对nintedanib 100 mg每日两次组BIBF 1202 ZW和BIBF 1202 glucuronide药代动力学参数的影响与150 mg每日两次组相似。
早餐剂量后1-1.6小时,午餐剂量后1-2小时,吡非尼酮的最大血浆浓度达到稳态,gMean为t1/2,ss约为3-4小时(表4)。血浆浓度和吡非尼酮的药代动力学参数没有受到与尼达尼布联合给药(表4)。
讨论
Ťhe results of this study show that nintedanib 150 mg twice daily had an acceptable safety and tolerability profile in Japanese patients with IPF when given alone or when added to ongoing pirfenidone therapy, but with more reports of nausea and vomiting when nintedanib was added to ongoing pirfenidone than when given alone. All the adverse events reported in this trial were mild or moderate in intensity. The most common adverse events reported in patients receiving nintedanib were gastrointestinal side-effects, which were reported by approximately one-third of patients taking nintedanib alone or nintedanib in addition to already prescribed pirfenidone therapy. Nausea, vomiting and diarrhoea were reported only by patients in the nintedanib 150 mg twice daily group: five (20.8%) patients on this dose reported nausea, five (20.8%) patients reported vomiting and four (16.7%) patients reported diarrhoea. Gastrointestinal adverse events were not unexpected, as gastrointestinal side-effects have been observed in previous trials of nintedanib (at doses up to 150 mg twice daily) and pirfenidone (at doses up to 3600 mg·day-1)在IPF患者[6-9,16],以及nintedanib在I期和II期的研究中,对于晚期实体肿瘤和晚期非小细胞肺癌患者,每天一次50毫克至300毫克,每天两次[17,18]。在本研究中,nintedanib 150 mg每日2次联合吡非尼酮治疗的患者中,69.2%报告了不良事件,而nintedanib 150 mg每日2次单独治疗的患者中,54.5%报告了不良事件。的差异主要是由于恶心(由四个(30.8%)病人nintedanib 150毫克每日两次除了pirfenidone和一个(9.1%)病人单独给nintedanib 150毫克每日两次)和呕吐(报道5(38.5%)的患者给予nintedanib 150毫克每日两次除了pirfenidone并没有单独nintedanib);除了吡非尼酮外,每天服用两次nintedanib 150毫克并不比单独服用更常见。1例患者出现呕吐,导致停药(除吡非尼酮每日3次外,nintedanib 150 mg每日2次,另加吡非尼酮200 mg每日3次)。与吡非尼酮(但非尼替丹)相关的其他不良事件包括皮肤光敏性和皮疹的剂量相关增加[8,11,12]。这些不良事件的发生率时,吡非尼酮与尼达尼布在这项研究共投并没有增加。
转氨酶水平的升高导致3名患者停药(所有患者每天两次服用nintedanib 150 mg)。转氨酶升高是可逆的,停药后升高消失。在一项针对日本晚期实体肿瘤患者的I期研究中,3名(25%)每天两次服用nintedanib 200 mg的患者的肝酶升高导致治疗中断[19]。在明天对IPF患者的II期试验中,有6名(7.1%)患者接受了每天两次的nintedanib 150 mg治疗,其肝脏酶水平出现了显著升高[6];这些升高是可逆的。在III期临床试验中,研究人员被建议通过中断治疗、减少剂量或停止治疗来控制肝酶升高。在这两项试验中,在nintedanib组中,5.0%的患者肝脏转氨酶升高(是天门冬氨酸转氨酶或丙氨酸转氨酶正常上限的三倍或更多),而在安慰剂组中,升高了0.7%。一般来说,在治疗中断、剂量减少或治疗中断后,肝酶水平恢复正常。用nintedanib治疗的患者中没有Hy’s law的病例[7]。
本研究在评价nintedanib的安全性和耐受性方面的一个局限性是其持续时间相对较短。除吡非尼酮外,nintedanib的长期安全性和耐受性的延伸试验正在进行中(www.clinicaltrials.gov标识符号码NCT01417156)。
尼达尼布的药动学特征之前已经在日本和白人患者描述的晚期实体瘤[18,19]。这些研究表明,有在日本和白人患者之间尼达尼布的药代动力学行为没有区别。尼达尼布的药代动力学分析显示适度快的吸收与适于一次或每天两次给药的终末半衰期。中号aximum plasma concentrations and exposure increased with doses from 50 mg once daily to 300 mg twice daily, both after single administration and at a steady state [17]。以前的报告显示所有的药代动力学变量显示中等高可变性,如预期的口服化合物[17,19]。在本研究中,对于晚期实体肿瘤患者,在每日两次给药nintedanib 150 mg后,tmax和半衰期的值与每日两次给药nintedanib 150 mg组的值相似。在这项研究中,曝光(最大浓度和AUCτss) nintedanib及其代谢物的往往是低nintedanib时添加到正在进行的pirfenidone疗法比单独时;但是,各个值的分布是重叠的。当在吡非尼酮治疗中加入nintedanib时,nintedanib的CL/F、ss和Vz/F、ss的值趋于升高,这表明联合使用吡非尼酮可能会降低nintedanib的生物利用度。由于两种药物的代谢途径不同,吡非尼酮与nintedanib合用时nintedanib的生物利用度较低,可能反映了吸收的减少。尼坦对吡非尼酮的药动学无影响。
总之,需要进一步研究的时候加入到吡非尼酮在IPF患者评估尼达尼布的安全性和耐受性。有对尼达尼布当尼达尼布加入正在进行的吡非尼酮的治疗及其代谢产物降低曝光的趋势。联合政府与尼达尼布对吡非尼酮的药代动力学无影响。吡非尼酮/尼达尼布方案的功效可能会在将来进行调查。
确认
病历书写援助,由勃林格殷格翰公司提供资金支持,是该手稿的制备过程中由伊丽莎白Ng和温迪·莫里斯福莱集团有限责任公司,中提供。提交人的所有内容和编辑决定全面负责,参与在稿件发展的各个阶段,并通过了最终版本。
脚注
有关编辑意见,请参阅欧元和J2015年;45:1208至1210年[10.1183/09031936.00043915]。
临床试验:这项研究是在注册www.clinicaltrials.gov标识号NCT01136174。
支持声明:本研究由勃林格殷格翰(Boehringer Ingelheim)资助。
利益冲突:可以在本文的网络版旁边找到披露信息www.qdcxjkg.com
- 收到了2013年11月13日。
- 公认2014年10月22日。
- 版权©2015人队