抽象
Nitazoxanide is widely available and exerts broad-spectrum antiviral activity体外。但是,没有证据表明它对SARS-COV-2感染的影响。
在多中心,随机的,双盲,安慰剂对照试验中,招收到Covid-19症状发病后长达3天的成年患者进行注册。在鼻咽拭子上通过RT-PCR确认SARS-COV2感染后,患者随机1:1接受尼硝唑(500mg)或安慰剂,TID,5天。主要结果是完全解决症状。二次结果是病毒载荷,实验室测试,炎症的血清生物标志物和住院率。还评估了不良事件。
从6月8日至8月20日起,筛查了1575名患者。其中,分析了392(198个安慰剂,194个硝唑)。从症状发作到第一剂研究药物的中位时间为5(4-5)天。在5天的学习访问中,北唑烷和安慰剂臂之间的症状分辨率没有差异。收集的拭子为硝唑酮臂中的29.9%的患者的SARS-COV-2负与18.2%的对照组(p = 0.009)。Viral load was also reduced after nitazoxanide compared to placebo (p=0.006). The percent viral load reduction from onset to end of therapy was higher with nitazoxanide (55%) than placebo (45%) (p=0.013). Other secondary outcomes were not significantly different. No serious adverse events were observed.
In patients with mild Covid-19, symptom resolution did not differ between nitazoxanide and placebo groups after 5 days of therapy. However, early nitazoxanide therapy was safe and reduced viral load significantly.
抽象
这是第一项评估尼唑烷治疗在轻度Covid-19中的效果的研究。奈唑纳尼德在治疗5天后没有加速症状分辨率,但没有明显减少病毒载荷,没有严重的不良事件。
介绍
The majority of patients infected with the severe acute respiratory syndrome coronavirus-2 (SARS-CoV-2) present mild symptoms of coronavirus disease 2019 (Covid-19) and recover with supportive care; however, in certain hosts, even mild disease may progress to clinical deterioration or a protracted course [1]。To date, no therapeutic interventions have proven effective in mild Covid-19.
在寻找有效的Covid-19疗法中,药物重估已被认为是一个重要的工具,因为它降低了开发成本和时间表[2]。已发现奈唑烷,临床批准的和市售的抗披物药物具有广谱抗病毒活性,包括针对冠状病毒,流感病毒和乙型肝炎和C病毒[3]。Furthermore, it has been shown to inhibit SARS-CoV-2 replication at low micromolar concentrations in Vero CCL81 cells [4]。此外,硝唑烷是口服生物可利用和广泛耐受的,从而代表Covid-19管理的有希望的替代方案是为了证明有效体内[3]。Efficacy in early treatment—initiated soon after symptom onset to reduce viral load and prevent disease progression—would be particularly appealing from a public health standpoint. However, there is no evidence of its safety or efficacy as therapy for mild Covid-19 patients.
在这种情况下,进行多中心,随机的安慰剂对照试验,以评估早期的硝唑烷治疗是否有效地加速症状分辨率。还评估了与安慰剂相比,还评估了病毒载荷,炎症,住院率和奈唑的安全性和奈唑烷的安全性。
Methods
学习规划
A double-blind, placebo-controlled trial was conducted at 5 freestanding urgent care centers and 2 hospitals across Brazil (supplementary table S1)。该审判由执行委员会设计,并由巴西国家研究伦理委员会批准(CAAE:32258920.0.1001.5257)和参与地点的个人伦理委员会。该试验由巴西科学,技术和创新部资助viathe National Council for Scientific and Technological Development (CNPq). Nitazoxanide was provided free of charge by Eurofarma, which had no further role in the design or conduct of the trial. The executive committee assures the accuracy of the data and fidelity of the trial to the protocol, which was registered in the Brazilian Registry of Clinical Trials (REBEC) number RBR-4nr86m and ClinicalTrials.gov numbernct04552483.。The independent Data and Safety Monitoring Board (DSMB), composed of experts in clinical trials and infectious diseases, was convened after 25%, 50%, and 75% of the participants had completed 14 days of follow-up and had access to information on adverse events and efficacy outcomes at every quartile.
The trial was conducted in accordance with the principles of the Declaration of Helsinki and the International Conference on Harmonisation (ICH) Good Clinical Practice Guideline (E6R2). Online clinical monitoring and quality control were outsourced to a contract research organisation (ATCGen, Campinas, Brazil). This report follows the Consolidated Standards of Reporting Trials (CONSORT) guideline [5]。审判议定书的最终议定书,修订和变更以及最终的统计分析计划详述补充方法。
耐心
Consecutive adult patients (aged 18 years or older) who presented with clinical symptoms of Covid-19 (defined for the purposes of this trial as dry cough, fever, and/or fatigue) of no longer than 3 days’ duration were enrolled. The exclusion criteria were: negative reverse-transcriptase quantitative real-time polymerase chain reaction (RT-PCR) test for SARS-CoV-2 on an nasopharyngeal swab specimen; inability to swallow; preexisting conditions precluding the safe conduct of study procedures, including severe renal, heart, respiratory, liver, or autoimmune diseases, cancer in the last 5 years, or known allergy or hypersensitivity to nitazoxanide; therapy with nitazoxanide in the 30 days before presentation; and clinical suspicion of bacterial pneumonia or tuberculosis.
随机化和掩蔽
使用计算机生成的随机数列表随机分配(1:1),以接收安慰剂或硝基唑(500mg口服溶液,20mg·mL−1[25 mL], three times daily for 5 days), dispensed by the pharmacy of each study site. Placebo and nitazoxanide were color-matched to ensure that assessors were unaware of group allocation at all time points.
程序
在第0天(基线),评估患者的资格。从每位患者获得知情同意,或者如果患者无法从医疗保健代理商无法提供同意。然后收集鼻咽拭子进行RT-PCR测试。为了减轻任何偏见,所有RT-PCR分析都在巴西联邦Minas Gerais大学CT-Vacinas中集中加工;标本由商业快递师收集当天发送。测试SARS-COV-2阳性的患者(RT-PCR后1-2天获得)(fig. 1)通过电话联系,并要求返回to the health facility to which they had originally presented. Site investigators then performed a comprehensive physical examination and measured body temperature, heart and respiratory rates, blood pressure, and peripheral oxygen saturation. Ethnicity, current medications, and date of symptom onset were self-reported by patients. Blood was then drawn for measurement of complete blood count, C-reactive protein (CRP), and serum biomarkers of inflammation (interleukin [IL]-6, IL-8, IL-1β, tumor necrosis factor [TNF]-α, and interferon [IFN]-γ) (See补充方法for details of RT-PCR testing and biomarker assessment)。
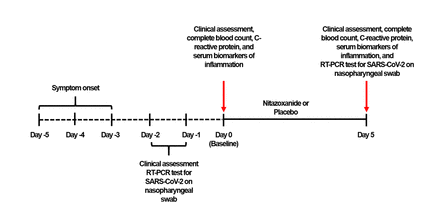
学习设计时间表。RT-PCR,实时逆转录聚合酶链反应。
All patients took home a symptom journal designed to gather information on daily symptoms, new symptoms, and the date of resolution of each symptom (supplementary table S2)。学习数据通过各学习网站的指定工作人员直接进入电子数据库,并通过ATCGEN的外部试验监测人员进一步验证。
One day after completion of therapy, patients returned to the study sites to return their symptom journals and provide a new nasopharyngeal sample for RT-PCR and blood samples for complete blood count, CRP, and serum biomarkers. Data validation was done exclusively by the designated study site manager, with assistance from ATCGen staff as needed.
Primary Outcome
在治疗5天后,主要结果完全解决了兴趣的三种症状(干咳,发热和/或疲劳)。
次要结果s
二次结果在鼻咽拭子上的病毒RNA载量降低了(从基线到治疗完成后的基线);改善实验室参数(包括炎症的血清生物标志物);治疗完成后医院入院的发病率。通过电话联系未返回研究部位或在治疗结束后接触治疗的患者,以了解非正常的原因,然后从每协定分析中排除(补充方法)。无论等级如何,通过审查通过第14天注册的电子医疗记录,身体检查,生命标志和实验室测试在整个审判中监测不良事件。使用监管活动®(Meddra,版本23.0)的医学词典进行分类。
统计分析
我们估计392名患者的样本大小(每只臂196,分配比例1:1)将提供85%的权力,以检测硝唑anide在α的双面意义水平的与安慰剂相比的症状无症状天数增加11%= 0.05。描述性统计数据用于人口统计学,实验室和临床数据。对于定性变量,费舍尔的确切测试进行了精确的测试。曼恩惠特尼Uand Wilcoxon tests were used for between and within-groups comparisons, respectively. Statistical analyses were performed in the SPSS Version 27 environment (IBM Corp, Armonk, NY), and a two-tailed p-value<0.05 was considered significant (补充方法)。
结果
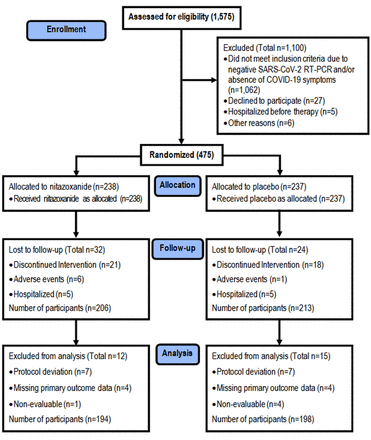
2020年6月8日至8月20日之间,评估了1575名患者在研究地点的资格。其中,475对SARS-COV-2感染的阳性进行RT-PCR和随机化。Figure 2总结了学习参与者的入学和后续行动。在随机化之前排除的原因包括在第0天收集的阴性RT-PCR和/或不存在Covid-19症状(n = 1062),拒绝参与(n = 27),在第一剂治疗前住院(n = 5)等等原因(n = 6)。在随机化(n = 475)后,由于中断的干预(n = 41),中度不良事件(n = 7)和住院(n = 10),患者被排除在外。六唑烷臂的六名患者和安慰剂臂中的一名患者由于中度腹泻而停止治疗,在前2天内呕吐;两者都经历了Covid-19症状的改善。由于临床恶化,10名患者(每只臂5个)都住院;没有完成5天的治疗过程。两名患者,均在硝唑烷臂,需要重症监护病房录取(supplementary table S3)。在分析期间,由于协议偏差,缺少关于主要结果的数据,或非评价性,12名患者从安慰剂臂中排除12名患者,导致392名患者的研究人群(194年在尼唑烷臂和198次在安慰剂手臂上)。
注册,随机化,随访和治疗。在研究部位评估了1575名患者的资格。其中,475对SARS-COV-2感染的阳性进行RT-PCR和随机化。在随机化之前排除的原因包括在第0天收集的阴性RT-PCR(基线)和/或缺乏Covid-19症状(n = 1062),拒绝参与(n = 27),在第一剂治疗前住院(n = 27)(n = 27)(n= 5)和其他原因(n = 6)。在随机化后(n = 475),由于中断的干预(n = 39),中度不良事件(n = 7)(所有胃肠镦折)和住院(n = 10)(每组5名患者,患者(每组5名患者,没有谁完成治疗)。在分析期间,由于协议偏差,缺少关于主要结果的数据,或非评价性,12名患者从安慰剂臂中排除12名患者,导致392名患者的研究人群(194年在尼唑烷臂和198次在安慰剂手臂上)。
Baseline characteristics
患者在基线时的特征表格1。人口统计学和临床特点alanced in both groups. Age ranged from 18 to 77 years. Just over half (53%) were women and 69% were white. Regarding timing of therapy initiation, 8% of patients were enrolled on day 1, 25% on day 2, and 67% on day 3 after symptom onset. The median (IQR) time between symptom onset and first dose of nitazoxanide or placebo was 5 (4–5) days. Fever, dry cough, and fatigue were the most frequent clinical features present at enrollment. At baseline, the overall median (IQR) viral load in the nasopharyngeal swab at baseline did not differ significantly between the two arms Additional characteristics of the study population at baseline are given insupplementary table S4。
Primary Outcome
完全解决症状(干咳,发烧和疲劳)在疗法5天后硝唑和安慰剂臂之间没有差异(fig. 3,supplementary figure S2)。
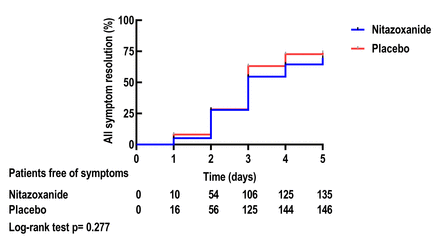
Kaplan-Meier治疗5天后症状分辨率曲线。
次要结果
After 5 days of therapy, viral load was lower in the nitazoxanide arm compared to placebo (p=0.006) (表3)。此外,来自第0天(基线)的病毒载量减少的百分比(表格1)至第5天(表2.) of therapy was significantly higher in the nitazoxanide arm (55%) than in the placebo arm (45%) (p=0.013). At the end-of-therapy visit, 29.9% of patients in the nitazoxanide arm与安慰剂臂18.2%在RT-PCR上的SARS-COV-2阴性(P = 0.009)(表3)。Vital signs (supplementary table S3), total leukocyte, neutrophil, lymphocyte, and platelet counts, CRP levels, and serum biomarkers of inflammation (表3)在硝唑烷酮臂的基线和第5天之间没有差异,奈唑烷和安慰剂臂之间也不差异。没有完成5天奈唑烷的患者住院治疗(supplementary table S3)。
任何手臂都没有报告任何死亡或危及生命的不良事件。在5天的治疗过程中,患者(奈唑烷,30.9%;安慰剂,30.4%)体验轻度和中度不良事件(表4)。最常见的不良事件(等级1和2级)是腹泻,头痛和恶心,群体之间没有显着差异。四名患者报告严重不良事件(单独头痛或腹泻);然而,它们在安慰剂臂中更频繁。变色的尿液是硝唑烷治疗的已知副作用,这可能会引发关于无意的解除盲目的担忧。然而,虽然这种事件在治疗臂中的频繁更频繁,但接受安慰剂的患者也报告了(5.6%)与1.5%,分别)(表3)。
讨论
在这种多中心,双盲,随机,安慰剂对照试验中的轻度Covid-19,我们发现症状分辨率(干咳,发热和疲劳)在治疗5天后尼硝唑和安慰剂之间没有差异。奈唑烷是安全的,病毒载荷显着降低,并增加了与安慰剂5天后的SARS-COV-2测试阴性的患者的比例。尼唑诺纳未防止住院或影响完整血统计数,CRP水平或血清生物标志物的任何变化。此外,尼硝唑烷是便宜的,广泛的可用性,并且迄今为止,没有证明其他推荐的疗法在该群体中提供任何益处。
迫切需要Covid-19的循证药物治疗方法。在大流行的挑战背景下,药物重估可以减少消耗耗时的药物开发过程,并迅速将有效疗法迅速部署到人口[2]。在实施本试验之前,NIH临床收集(NCC)图书馆,筛选了727名FDA批准的药物或药物样化合物,具有人类临床试验的使用历史,用于潜力体外antiviral activity against SARS-CoV-2 (补充图S1)。Nitazoxanide and tizoxanide, its active metabolite, significantly reduced viral load in VeroE6, human embryonic kidney (HEK 293T), and lung epithelial (Calu-3) cells infected with SARS-CoV-2, without inducing loss of cell viability. The adverse effect profile of nitazoxanide is well-known, since it has been commercially available and used in the clinic since 1996; indeed, a commercial formulation was used in this trial. A dosage regimen of 500 mg every 12 h is approved and commonly prescribed for treatment of intestinal parasitosis [5],少数报告不良事件。在我们的研究中,施用相同的剂量,但每8小时,基于体外pharmacological studies by our group and published data on plasma concentration [6–8],为了最大化SARS-COV2的潜在抑制作用体内[7]。我们假设剂量在人体中的最大血清浓度的高比率在人类中到最大体外有效的抗病毒浓度将转化为较高的潜力,以在批准的剂量下实现病毒抑制[10]。
Nitazoxanide failed to meet the primary outcome in patients with mild Covid-19 when evaluated after 5 days of therapy. Consistent with the体外数据,我们观察到尼沙妥昔尼患者在温和Covid-19患者的5天后病毒载量的显着减少。这种效果可能具有流行病学影响,潜在地降低SARS-COV-2的社区传播[9,10],发病率[11], and mortality [12]。
我们在症状发作后尽快注册患有轻度Covid-19的患者,因为抗病毒在这种感染阶段似乎更有效[13]。The cutoff point for enrollment was 3 days after onset of first symptom; however, as therapy was begun only after confirmation of SARS-CoV-2 infection, the median timing of treatment initiation was 5 (4–5) days after symptom onset (表格1)。In previous trials of putative antiviral agents, median time to initiation of treatment was 13 days [14], 9 days [15],8-9天[16], and 4–5 days [17]症状后。即使早期的启动治疗也可能更有效,因为SARS-COV-2病毒载量通常在症状发作之前发生[18] and systemic hyperinflammation rather than viral pathogenicity dominates later stages of Covid-19, at which point antiviral therapy could be ineffective [18,19]。Additionally, analyses were done 5 days after therapy since a longer period of time would result in symptom resolution in most patients independent of treatment; in previous studies, the median to time from onset to resolution of symptoms has been reported as 8 (6.25–11.5) days [20]。
Besides timing, two additional points which may have biased our findings in favor of the nitazoxanide arm were the severity of Covid-19 at presentation and the favorable demographic profile of the sample. Only patients with mild Covid-19 were enrolled; most were young adults (aged 18–39), few had comorbidities (12–18%), and use of concomitant medications was infrequent (less than 20% of the sample). Further studies are needed to evaluate whether nitazoxanide may play a role in more advanced Covid-19. In this line, Hung等等。最近报道了将抗病毒疗法与严重Covid-19患者结合的重要性,旨在减少病毒载荷和减轻症状[17]。
硝唑烷的剂量频率的增加不会导致不良事件率的任何显着变化。除了尿液变色(已知和无害效果)之外,武器之间的频率和严重程度都不有显着不同,表明奈氮烷对轻度Covid-19患者安全。
This study has several limitations. Only three symptoms (dry cough, fever, and fatigue) were considered for analysis of primary outcome, even though Covid-19 is now known to have protean manifestations and patients reported other symptoms. Patients were followed during the 5-day course of therapy; however, a long-term analysis of the effects of therapy should be performed. All patients were instructed to take their study medication 3 times daily as prescribed, return to the study site if symptoms worsened or adverse effects developed, and complete their symptom journals with data on any adverse events, the intensity of each Covid-19 symptom, and when symptoms abated and resolved. However, we cannot ascertain the extent to which patients followed these instructions. Nevertheless, given the placebo-controlled design, nonadherence may have occurred in both groups. In the nitazoxanide group, around 3% stopped therapy due to gastrointestinal upset after Covid-19 symptoms had already improved. Finally, no chest computed tomography scans or plain radiographs were obtained in these patients, since our work was mostly performed at freestanding mobile urgent care centers.
In summary, in patients with mild Covid-19 enrolled within 3 days of symptom onset, nitazoxanide as compared with placebo was not an effective therapy in terms of accelerating symptom resolution after 5 days of therapy, and did not modify clinically relevant secondary outcomes. However, nitazoxanide was safe, significantly decreased viral load, and increased the proportion of patients who tested negative for SARS-CoV-2 after completion of therapy.
Acknowledgements
作者感谢MoiraElizabethSchöttler(Rio de Janeiro,巴西)和Filippe Vasconcellos(巴西圣保罗)进行编辑援助。
脚注
本文提供了补充材料www.qdcxjkg.com.
ClinicalTrials.gov numbernct04552483.
Data availability:合格的临床研究人员可以通过提交对通讯作者提出有价值的研究问题的提案,要求获得去识别的参与者数据集,知情同意书和相关文件,包括研究议定书,包括提交本条提出本条的研究议定书。应签署合同。
Data sharing:Qualified clinical researchers can request access to de-identified participant data set (PP and ITT), informed consent forms, and related documents including the study protocol that underlie this article through submission of a proposal with a valuable research question to the corresponding author. A contract should be signed.
支持声明:巴西科学委员会的支持and Technological Development (CNPq), Brazilian Ministry of Science, Technology, and Innovation for Virus Network; Brasília, Brazil, number: 403485/2020-7 and Funding Authority for Studies and Projects, Brasília, Brazil, number: 01.20.0003.00. Conselho Nacional de Desenvolvimento Científico e Tecnológico;DOI:http://dx.doi.org/10.13039/501100003593;格兰特:403485 / 2020-7。
Autor贡献者:PRMR,PLS,FFC负责手稿的设计,分析和写作。Macmj,PGGMMT,MAM,LFGO,CCL,EZS,WFJ,EM,NFM,JMJG,MNC,ISS,NFP,PVMM负责招募和临床护理。APSMF,KGF,RPR,AFC,PAA,JLM,ATC,DBBT,REM负责实验室分析。RRL,JRL和PP负责统计分析。KGF,JRL和PP也负责写作稿件。所有作者审查并批准了稿件的最终版本。
利益冲突:Rocco博士在提交的工作之外向Sanofi报告个人费用;
Conflict of interest: Dr. Silva has nothing to disclose.
Conflict of interest: Dr. Cruz has nothing to disclose.
Conflict of interest: Dr. Junior has nothing to disclose.
利益冲突:Tierno博士没有什么可披露的。
利益冲突:Moura博士无需披露。
Conflict of interest: Dr. Oliveira has nothing to disclose.
利益冲突:利马博士无需披露。
利益冲突:桑托斯博士无需披露。
Conflict of interest: Dr. Junior has nothing to disclose.
利益冲突:Fernandes博士无需披露。
兴趣冲突:弗兰卡尼博士没有披露。
兴趣冲突:Magri博士没有披露。
兴趣冲突:莫拉斯博士无需披露。
兴趣冲突:戈兰博士没有披露。
兴趣冲突:Carbonieri博士无需披露。
利益冲突:桑托斯博士无需披露。
利益冲突:PAES博士无需披露。
Conflict of interest: Dr. Maciel has nothing to disclose.
Conflict of interest: Dr. Rocha has nothing to disclose.
Conflict of interest: Dr. Carvalho has nothing to disclose.
利益冲突:Alves博士没有披露。
利益冲突:摩德纳博士无需披露。
Conflict of interest: Dr. Cordeiro has nothing to disclose.
Conflict of interest: Dr. Trivella has nothing to disclose.
Conflict of interest: Dr. Marques has nothing to disclose.
Conflict of interest: Dr. Luiz has nothing to disclose.
Conflict of interest: Dr. Pelosi has nothing to disclose.
Conflict of interest: Dr. Silva has nothing to disclose.
- 收到October 5, 2020.
- AcceptedDecember 4, 2020.
- 复制right ©ERS 2020
本文是开放式访问,并根据创意公约188滚球软件归因非商业许可证4.0的条款分发。