Abstract
ObjectiveTo evaluate pulmonary function and clinical symptoms in coronavirus disease 2019 (COVID-19) survivors within 3 months after hospital discharge, and to identify risk factors associated with impaired lung function.
Methods and materialCOVID-19 patients were prospectively followed-up with pulmonary function tests and clinical characteristics for 3 months following discharge from a hospital in Wuhan, China between January and February 2020.
Results647 patients were included. 87 (13%) patients presented with weakness, 63 (10%) with palpitations and 56 (9%) with dyspnoea. The prevalence of each of the three symptoms were markedly higher in severe patients than nonsevere patients (19%versus10% for weakness, p=0.003; 14%versus7% for palpitations, p=0.007; 12%versus7% for dyspnoea, p=0.014). Results of multivariable regression showed increased odds of ongoing symptoms among severe patients (OR 1.7, 95% CI 1.1–2.6; p=0.026) or patients with longer hospital stays (OR 1.03, 95% CI 1.00–1.05; p=0.041). Pulmonary function test results were available for 81 patients, including 41 nonsevere and 40 severe patients. In this subgroup, 44 (54%) patients manifested abnormal diffusing capacity of the lung for carbon monoxide (DLCO) (68% severeversus42% nonsevere patients, p=0.019). Chest computed tomography (CT) total severity score >10.5 (OR 10.4, 95% CI 2.5–44.1; p=0.001) on admission and acute respiratory distress syndrome (ARDS) (OR 4.6, 95% CI 1.4–15.5; p=0.014) were significantly associated with impairedDLCO.Pulmonary interstitial damage may be associated with abnormalDLCO.
ConclusionPulmonary function, particularlyDLCO, declined in COVID-19 survivors. This decrease was associated with total severity score of chest CT >10.5 and ARDS occurrence. Pulmonary interstitial damage might contribute to the impariedDLCO.
Abstract
COVID-19 patients present with impairedDLCOat 90 days after discharge, particularly severe patients. Chest CT TSS >10.5 and ARDS occurrence are associated with impairedDLCO.Pulmonary interstitial damage may contribute to the impairedDLCO.https://bit.ly/2JevUtm
Introduction
The epidemic of coronavirus disease 2019 (COVID-19) has had devastating effects. Patients surviving hospitalisation are frequently reported to have pulmonary sequelae. It is challenging to evaluate lung function throughout COVID-19 progression because of the difficulty related to infection control risks in obtaining lung function testing during this contagious pandemic disease [1].
There are short reports of lung function of COVID-19 patients at discharge and at 30 days post-discharge [2,3]. Reduced lung function was demonstrated in survivors of severe acute respiratory syndrome (SARS) and Middle East respiratory syndrome (MERS) up to 6 months following hospital discharge [4,5]. SARS patients were reported to have a mild decrease in diffusing capacity of the lung for carbon monoxide (DLCO) 6–8 weeks after discharge, with improving lung function over time [6]. We performed a prospective cohort study to identify main sequelae and lung function changes in hospitalised severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) patients during 3-month follow-up.
Methods
Study design
This was a prospective cohort study performed at the Affiliated Hospital of Jianghan University (Wuhan, China). COVID-19 was confirmed and diagnosed according to Chinese management guideline for COVID-19 [7]. Nucleic acid tests were provided for SARS-CoV-2 by real-time reverse-transcriptase PCR assays. Severity was defined by the World Health Organization guideline for COVID-19 [8]. Severe pneumonia refers to fever or suspected respiratory infection, plus one of respiratory rate >30 breaths·min−1, severe respiratory distress or peripheral oxygen saturation ≤93% on room air. A total of 749 COVID-19 patients with full data available were admitted from January to February 2020. Of those, 81 patients died within 3 months and 21 patients were lost during follow-up. 647 patients were followed-up for 3 months after discharge. Patients' clinical baseline details, major clinical characteristics and lung function within 3 months' follow-up were recorded. Ethics approval was obtained from the medical ethics committee of Jianghan University Affiliated Hospital and China–Japan Friendship Hospital (WHSHIRB-K-2020015). Before data collection, we obtained patients’ consent.
Pulmonary function tests
81 (13%) patients underwent pulmonary function testing (PFT) at 3 months after discharge. PFT was performed by a professional doctor with 20 years of experience using the MasterScreen PFT system (Jaeger, Hoechberg, Germany) at the 3-month follow-up visit. The recorded parameters are as follows: total lung volume (TLC), residual volume, forced vital capacity (FVC), forced expiratory volume in 1 s (FEV1), FEV1/FVC ratio, maximum mid-expiratory flow (MMEF) andDLCO.
Chest computed tomography visual quantitative evaluation
Chest computed tomography (CT) scans were performed on 16- or 64-multidetector CT scanners (GE LightSpeed 16; GE Healthcare, Wauwatosa, WI, USA or Somatom Sensation 64; Siemens Healthcare, Malvern, PA, USA). All patients underwent chest CT scan at admission. In addition, 45 out of 81 patients who had PFT had chest CT scan at 3 months after discharge. Two experienced radiologists (YL Wan and HB Li) reviewed CT images without knowledge of mild or severe, normal or reducedDLCO.Pulmonary interstitial changes on follow-up chest CT graph was defined as a combination of findings including fibrous stripe, ground-glass opacity, consolidation, subpleural curvilinear shadow, coarse reticular pattern and traction bronchiectasis [9]. Meanwhile, main pulmonary artery (MPA), ascending aorta (AAo) diameters and the MPA/AAo ratio were measured to evaluate the relationship between pulmonary vascular disease and impairedDLCO[10]. To explore the relationship between impairedDLCOand radiographic changes, we conducted chest CT total severity score (TSS), which was evaluated by percentage of involvement in each lobe and overall lung. The percentage of the lobar involvement in each of the five lung lobes were classified in five levels: 0 (0%), 1 (1–25%), 2 (26–50%), 3 (51–75%) and 4 (76–100%). The TSS was obtained by adding the five lobar scores [11].
Statistical analysis
Data were expressed as number (%) for categorical variables, mean±sdwhen they were normally distributed and median (interquartile range (IQR)) when they had skewed distributions. t-test, Mann–Whitney U-test, Chi-squared test or Fisher's exact test were used to compare differences in characteristics and pulmonary function between groups of different characteristics. We used univariable and multivariable logistic regression models to explore the risk factors associated with the occurrence of sequelae and impaired lung function. To avoid overfitting in the multivariable logistic regression model, we chose just four variables for analysis considering the total number of impairedDLCO(n=41) in our study. The receiver operating characteristic (ROC) curve and the area under the curve were used to assess the predicted value of TSS for impairedDLCO.All statistical analyses were performed using SPSS (version 24; IBM, Armonk, NY, USA) and Prism (version 8.0.1; GraphPad, San Diego, CA, USA) with two-tailed p<0.05.
Results
Patient characteristics
647 COVID-19 patients attended the 3-month follow-up visit after hospital discharge, including 399 nonsevere patients and 248 severe patients (supplementary figure S1). The mean±sdage was 58±15 years, with 44% being male. The sequential organ failure assessment (SOFA) score on admission was 1 (IQR 0–2). The top three comorbidities were hypertension (30%), diabetes (11%) and chronic respiratory disease (6%). Moreover, severe patients were significantly older and had higher SOFA score at admission (supplementary table S1).
The residual symptoms at 3-month follow-up
Ongoing symptoms for COVID-19 at the 3-month follow-up visit were weakness, palpitations, dyspnoea, cough, lower limb oedema, chest pain and haemoptysis. 87 (13%) patients presented with fatigue in their daily lives, 63 (10%) with palpitations and 56 (9%) with dyspnoea. Prevalences of the three symptoms in patients with severe COVID-19 were markedly higher than those in the nonsevere patients (19%versus10% for weakness, p=0.003; 14%versus7% for palpitation, p=0.007; 12%versus7% for dyspnoea, p=0.014) (supplementary table S1). Insupplementary table S2, we compare the clinical characteristics between COVID-19 patients who had sequelae or not. The main differences between these two groups were disease severity, inpatient days, inflammation and coagulant disorder on admission. In univariable analysis, odds of sequelae were significantly higher in severe patients with long hospital stay. In addition, higher white blood cell count, higher hypersensitive C-reactive protein and abnormal D-dimer levels were associated with the occurrence of sequelae. In the multivariable model, severity and inpatient days were significantly associated with the occurrence of sequelae (supplementary table S3).
Lung function at 3-month follow-up
81 COVID-19 patients were assessed for pulmonary function test at 3 months after discharge, including 41 nonsevere and 40 severe patients. In this subgroup of the cohort, the mean±sdage was 59±14 years, 34 (42%) patients were male and the mean±sdbody mass index was 23.87±3.18 kg·m−2.As shown intable 1, pulmonary function was impaired in 61 (75%) out of 81 survivors. Eight (10%) patients had reduced TLC; 17 (21%) patients had decreased FVC; in five (6%) patients, FEV1/FVC ratio was <70%; and in 44 (54%) patients,DLCO< 80%的预测。以确定是否异常lung function was associated with disease severity, we compared characteristics and pulmonary function parameters between nonsevere and severe cases. There were no significant differences in TLC, FVC, FEV1, FEV1/FVC, MMEF at 25–75% of FVC orDLCOto alveolar volume ratio, according to the spirometry between nonsevere and severe patients. However, significant difference was found forDLCO,which was <80% pred for 68% of severe patients compared to 42% of nonsevere patients (p<0.05) (figure 1).
Lung function in severe and nonsevere coronavirus disease 2019 patients
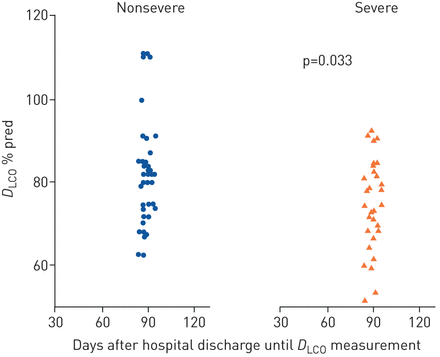
Percentage of predicted diffusing capacity of the lung for carbon monoxide (DLCO) at 3 months after discharge in nonsevere and severe coronavirus disease 2019 patients.
44 out of 81 COVID-19 patients had impairedDLCO.To figure out the differences between normal and impairedDLCOpatients, we compared clinical characteristics between two groups intable 2.We found that parameters including severity, chest CT TSS, lymphocyte count, MPA diameter on admission and acute respiratory distress syndrome (ARDS) were higher in patients with impairedDLCO统计学的,两组之间的差异tically significant.
Characteristics in coronavirus disease 2019 patients with normal and impaired diffusing capacity of the lung for carbon monoxide (DLCO) at 3-month follow-up
TSS of chest CT, pulmonary interstitial damage, vascular disease and impairedDLCO
All patients underwent chest CT scan at admission; in 90% of patients, lesions could be seen in the bilateral lung on admission and there was no statistical difference between the normal and impairedDLCOgroups (p=0.459). To evaluate the effect of CT assessment onDLCOdecline in patients with COVID-19, we calculated CT TSS for all patients. The median (IQR) TSS was 9 (5–13) at admission. We created a ROC curve to explore cut-off of TSS to predict abnormalDLCO.We found that the cut-off of TSS was 10.5 on admission; the area under the curve was 0.765 (95% CI 0.663–0.867; p<0.001), with sensitivity 64% (95% CI 49–76%) and specificity 84% (95% CI 69–92%) (figure 2).
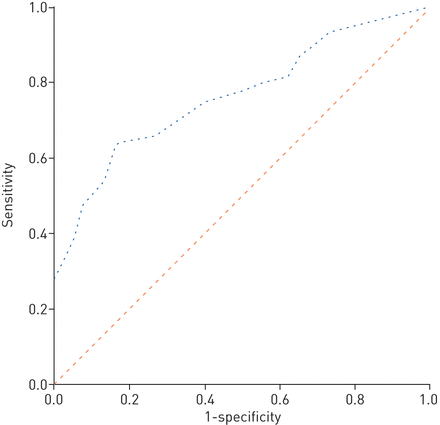
Receiver operating characteristic curve analysis of total computed tomography severity score (TSS) on admission for prediction of impaired diffusing capacity of the lung for carbon monoxide during 3-month follow-up. With the cut-off value of 10.5 for the TSS, the area under the curve was 0.765 (95% CI 0.663–0.867; p<0.001) with sensitivity 64% and specificity 84%.
45 out of 81 patients who underwent PFT had a chest CT scan at 3-month follow-up after discharge. To determine whether pulmonary interstitial damage contributed to impairedDLCOor not, we analysed pulmonary CT changes at 3 months after discharge. It was found that patients with abnormalDLCOwere more likely to have interstitial damage, especially manifesting the signs of traction bronchiectasis, subpleural curvilinear shadow and coarse reticular pattern, indicating that pulmonary interstitial damage may contribute to impairedDLCOin COVID-19 patients (table 3). When exploring the relationship between vascular disease and impairedDLCO, no significant differences were found at 3 months after discharge.
Pulmonary computed tomography scan and impaired diffusing capacity of the lung for carbon monoxide (DLCO) at 3 months
Predictors for lung function decline
Univariable logistic analysis showed that severity, TSS >10.5, MPA diameter at admission and ARDS were significantly associated with impairedDLCO.Other variables were not associated withDLCOdecline. Finally, we put age, MPA diameter, TSS >10.5 based on the ROC curve and ARDS into the multivariable logistic regression model. Details of univariable and multivariable analyses are shown insupplementary table S4andfigure 3.We found that TSS >10.5 (OR 10.4, 95% CI 2.5–44.1; p=0.001) and ARDS (OR 4.6, 95% CI 1.4–15.5; p=0.014) were significantly associated with impairedDLCO.
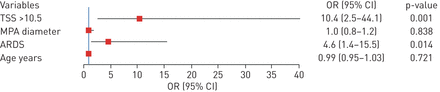
Factors associated with impaired diffusing capacity of the lung for carbon monoxide (DLCO在多变量回归三个月随访sion analysis. TSS: total severity score; MPA: main pulmonary artery; ARDS: acute respiratory distress syndrome.
Discussion
在目前的研究中,我们专注于研究the residual symptoms and pulmonary functions in COVID-19 patients after hospital discharge. Our study revealed that the most common residual symptoms were weakness (13%), palpitations (10%) and dyspnoea (9%). Prevalences of these three symptoms were significantly higher in severe COVID-19 patients than that in nonsevere patients. In addition, COVID-19 patients presented with abnormal pulmonary function, especially impairedDLCOduring recovery. Furthermore, patients with severe COVID-19 were found to have a higher prevalence of impairedDLCO.Finally, multivariable analysis in our study demonstrated that TSS >10.5 and ARDS were significantly associated with impairedDLCO.Pulmonary interstitial damage may contribute to impairedDLCOat 3 months after discharge.
During the initial epidemic of COVID-19, the most common symptoms at the onset of illness were fever, cough, fatigue and shortness of breath [12,13]. Some symptoms may continue following hospital discharge. It has been reported that weakness is common after acute lung injury and is associated with substantial impairments in physical function and quality of life [14]. The potential cause of these sequelae was multiple organ injury following SARS-CoV-2 infection. A prior study has shown that COVID-19 patients can have impaired physical functioning when discharged home, even after early physiotherapy [15]. For patients with SARS and MERS, the 6-min walk distance was reduced at 3 months after hospital discharge, but could be slowly improved by 12 months [16,17]. Fatigue was reported for at least one-third of patients when followed-up for 18 months [18] and 40 months [19]. In this study, 10% patients experienced palpitations. It was well reported that myocardial injury was common in COVID-19 patients. According to autopsy findings, viraemia in six out of 10 and five out of 12 patients demonstrated high viral RNA titres in the liver, kidney or heart [20]. In addition, myocardial injury was highly associated with fatal outcomes in this infectious disease [21,22]. Different degrees of myocardial injury might be a reason for palpitations.
Reports by Moet al.[3] and Huanget al.[2] showedDLCOdysfunction in COVID-19 patients at discharge and 1 month from discharge, respectively. According to their studies, anomalies were noted inDLCO% pred in 47.2% and 52.6% of patients, respectively. They all reported the significant difference in impaired diffusing capacity among the different groups of severity. In our study, 44 (54%) patients had impaired diffusing capacity and there was a significant difference between nonsevere and severe COVID-19 patients at 3-month follow-up, which is in agreement with previous studies. Lung function disorder is one of the common issues with SARS and MERS patients. Pulmonary function defects were detected in half of the recovered SARS patients 3 months after hospital discharge [23]. Interstitial or pulmonary vascular abnormalities are associated with reducedDLCO[24], but it is unclear whether impairedDLCOin COVID-19 is due to pulmonary interstitial or pulmonary vascular disease, or both. In order to illuminate the reason for impairedDLCOin COVID-19, we analysed the TSS score on admission, pulmonary interstitial abnormalities at 3 months after discharge and markers of vasculopathy (including D-dimer, Padua score, MPA diameter, MPA/AAo ratio and use of low molecular weight heparin) and explored their associations withDLCOdecline. As a method to score the severity of inflammation on CT images [11], TSS >10.5 was found to be significantly associated with impairedDLCO, indicating that the severity of pulmonary inflammation may be the reason for impairedDLCO.结果暗示我们应该跟进COVID-19 patients for pulmonary function, especially those individuals with high TSS upon chest CT. Additionally, we found that patients with impairedDLCOhad a higher percentage of interstitial lesions, indicating that pulmonary interstitial damage may contribute to impairedDLCOat 3 months after discharge. There was no significant difference in vascular diseases between impaired and normalDLCOat 3-month follow-up. However, due to small sample size and lack of CT pulmonary angiography, the results could not accurately reflect the relationship between vascular abnormalities and impairedDLCO.The pathogenesis of impairedDLCOin COVID-19 merits further study in the future.
We analysed inherent relationships of corticosteroids treatment, inflammation on admission and ARDS occurrence with impairedDLCO.As a result, ARDS occurrence contributed to impairedDLCO, which was consistent with previous reports that ARDS survivors had striking decline inDLCO, the most common abnormality in pulmonary function [25,26]. The exact pathologic causes of lung dysfunction in recovered COVID-19 patients remain unknown. Structural pulmonary damage caused by the ARDS and subsequent chronic changes may damage gas exchange [27]. Furthermore, neuromuscular weakness may also contribute to the impaired pulmonary function [28].
尽管大量的患者随访in our cohort, there were several limitations to our study. Firstly, pulmonary function testing was not carried out for all patients and not all patients undergoing lung function test received a chest CT scan at 3-month follow-up, mainly attributed to our limited knowledge of this novel virus and poor awareness about its impact on patients' lung function in the early epidemic era. This is the inherent limitation of this real-world study. Secondly, there was a lack of CT pulmonary angiography and other instruments to evaluate cardiovascular conditions. Furthermore, there was no direct evidence to explain the aetiology of sequelae and impairedDLCOin COVID-19 survivors. Although psychiatric and traumatic stress disorders were reported for patients with SARS and MERS [29,30], a larger study with long-term follow-up needs to be carried out.
Conclusion
Weakness, palpitations and dyspnoea were the most common sequelae of COVID-19. Lung carbon monoxide diffusion dysfunction was the major damage in pulmonary function of COVID-19 survivors at 3 months after discharge. Chest CT TSS >10.5 and ARDS occurrence in COVID-19 were associated with impairedDLCO.Pulmonary interstitial damage may contribute to impairedDLCOat 3 months after discharge. This indicates that there is a necessity to adopt pulmonary rehabilitation strategy to improve outcomes in COVID-19 patients.
Supplementary material
Supplementary Material
Please note:supplementary material is not edited by the Editorial Office, and is uploaded as it has been supplied by the author.
Supplementary materialERJ-03677-2020.SUPPLEMENT
Shareable PDF
Supplementary Material
This one-page PDF can be shared freely online.
Shareable PDFERJ-03677-2020.Shareable
Acknowledgements
We thank Min Liu, Ziming Wang, Di Xu and Wei Yu (Affiliated Hospital of Jianghan University, Wuhan, China), for their data collection. We also thank Bruce L. Davidson, from the Division of Pulmonary and Critical Care Medicine, Providence Health System (Seattle, WA, USA), for his suggestions and language editing. They were not compensated for their contributions.
Footnotes
This article has supplementary material available fromwww.qdcxjkg.com
Author contributions: W. Qin, S. Chen, Y. Zhang, B. Hu, Z. Zhang and C. Li conceived the study. W. Qin, B. Hu, S. Chen, Z. Zhu, F. Li, X. Wang, Y. Zhang, Y. Wang, K. Zhen, J. Wang, Y. Wan and H. Li collected data. W. Qin, F. Dong, Z. Zhang, I. Elalamy, C. Li and Z. Zhai analysed and interpreted data. W. Qin, F. Dong, Y. Zhang and S. Chen drafted the manuscript. I. Elalamy, C. Li, Z. Zhai and C. Wang revised the manuscript. Z. Zhai, C. Li and C. Wang obtained funding and supervised the study.
Conflict of interest: W. Qin has nothing to disclose.
Conflict of interest: S. Chen has nothing to disclose.
Conflict of interest: Y. Zhang has nothing to disclose.
Conflict of interest: F. Dong has nothing to disclose.
Conflict of interest: Z. Zhang has nothing to disclose.
Conflict of interest: B. Hu has nothing to disclose.
Conflict of interest: Z. Zhu has nothing to disclose.
Conflict of interest: F. Li has nothing to disclose.
Conflict of interest: X. Wang has nothing to disclose.
Conflict of interest: Y. Wang has nothing to disclose.
Conflict of interest: K. Zhen has nothing to disclose.
Conflict of interest: J. Wang has nothing to disclose.
Conflict of interest: Y. Wan has nothing to disclose.
Conflict of interest: H. Li has nothing to disclose.
Conflict of interest: I. Elalamy has nothing to disclose.
Conflict of interest: C. Li has nothing to disclose.
Conflict of interest: Z. Zhai has nothing to disclose.
Conflict of interest: C. Wang has nothing to disclose.
Support statement: This study was supported by a grant from the Chinese Academy of Engineering emergency research and cultivation project for COVID-19 (2020-KYGG-01-05), National Key Research and Development Program of China (number 2016YFC0905600; 2016YFC0901104; 2018YFC1315100), CAMS Innovation Fund for Medical Sciences (CIFMS) (number 2018-I2M-1-003) and National Natural Science Foundation of China (number 81570049; 81970058). The funding bodies are not involved in the design of the study, collection, analysis, interpretation of data or in writing the manuscript. Funding information for this article has been deposited with theCrossref Funder Registry.
- ReceivedSeptember 29, 2020.
- AcceptedDecember 15, 2020.
- Copyright ©ERS 2021
This version is distributed under the terms of the Creative Commons Attribution Non-Commercial Licence 4.0.















