Abstract
BackgroundPulmonary arterial hypertension (PAH) is a progressive disease characterised by pro-proliferative and anti-apoptotic phenotype in vascular cells, leading to pulmonary vascular remodelling and right heart failure. Peptidyl-prolylcis/transisomerase, NIMA interacting 1 (Pin1), a highly conserved enzyme, which binds to and catalyses the isomerisation of specific phosphorylated Ser/Thr-Pro motifs, acts as a molecular switch in multiple coordinated cellular processes. We hypothesised that Pin1 plays a substantial role in PAH, and its inhibition with a natural organic compound, Juglone, would reverse experimental pulmonary hypertension.
ResultsWe demonstrated that the expression of Pin1 was markedly elevated in experimental pulmonary hypertension (i.e.hypoxia-induced mouse and Sugen/hypoxia-induced rat models) and pulmonary arterial smooth muscle cells of patients with clinical PAH.In vitroPin1 inhibition by either Juglone treatment or short interfering RNA knockdown resulted in an induction of apoptosis and decrease in proliferation of human pulmonary vascular cells. Stimulation with growth factors induced Pin1 expression, while its inhibition reduced the activity of numerous PAH-related transcription factors, such as hypoxia-inducible factor (HIF)-α and signal transducer and activator of transcription (STAT). Juglone administration lowered pulmonary vascular resistance, enhanced right ventribular function, improved pulmonary vascular and cardiac remodelling in the Sugen/hypoxia rat model of PAH and the chronic hypoxia-induced pulmonary hypertension model in mice.
Conclusion我们的研究的发展rates that targeting of Pin1 with small molecule inhibitor, Juglone, might be an attractive future therapeutic strategy for PAH and right heart disease secondary to PAH.
Abstract
Peptidyl-prolylcis/transisomerase, NIMA interacting 1 (Pin1) enzyme inhibition by Juglone administration reversed both hypoxia- and non-hypoxia-driven experimental PAH by improving pulmonary vascular remodelling and right ventricular functionhttps://bit.ly/3zWqTvb
Introduction
Pulmonary arterial hypertension (PAH) is a fatal pulmonary vascular disease characterised by pulmonary vascular remodelling and increased pulmonary vascular resistance, culminating in right ventricular (RV) hypertrophy and failure [1]。The dynamic vasoconstriction of pulmonary arteries, their adverse structural remodelling, fibrosis and stiffening, are the main causes of increased pulmonary vascular resistance [2]。PAH shares some common features with cancer, such as the increase in cell proliferation and resistance to apoptosis [3]。除此之外,内皮功能障碍,一个分辨ase in inflammation, dysregulated angiogenesis, metabolic disturbance, oxidative stress and mitochondrial dysregulation contribute to the progression of the disease [4]。PAH is a complex and multifactorial disease, the emergence of which has been explained by a concept known as the “multiple-hit hypothesis”, in which a combination of two or more hits is essential for triggering of key mechanisms that lead to vascular constriction [5–7]。
The peptidyl-prolylcis/transisomerase, NIMA interacting 1 (Pin1), is a highly conserved enzyme, which belongs to the PPI-ase superfamily of proteins, comprising of cyclophilins, FK506-binding proteins (FKBPs) and parvulins [8]。Pin1 is the only known PPI-ase, which specifically and uniquely recognises phosphorylated Ser/Thr-Pro peptides and catalyses thecis/transisomerisation of these pSer/Thr-Pro motifs [9,10]。Post-phosphorylationcis/transisomerisation, driven by Pin1, induces structural changes further affecting the phosphorylation/dephosphorylation status of target proteins [11–14]。As a consequence, conformational modifications of many target phosphoproteins affect their stability and activity, subcellular localisation and protein–protein interactions, triggering multiple cellular signalling pathways [14–16]。Thus, Pin1 serves as a molecular determinant of the fate of phosphoproteins [14], which adds a new layer of control in various signalling pathways [17]。Among Pin1 substrates are numerous cell cycle-regulatory proteins, such as cell division cycle 25 [18], cyclin D1 [19,20], cyclin E [21] and polo-like kinase 1 [22,23]; transcription factors, such as retinoblastoma protein [24] and neurogenic locus notch homologue protein 1 (Notch1) [24]; DNA-damage factors, such as CREB-binding protein/p300 [25] and homeodomain-interacting protein kinase 2; and apoptotic regulatory proteins, such as tumour suppressor p53 [26,27] and survivin [28]。In association with this, a number of signalling pathways are markedly affected by Pin1, including cell cycle progression, gene transcription, tumour development, oxidative stress and apoptosis [17]; thus, regulation driven by Pin1 provides a new platform for assembly of multiple protein networks.
Here we investigated the expression and the role of Pin1 in experimental and clinical PAH by using Juglone, a specific and irreversible Pin1 inhibitor, established to covalently inactivate Pin1 [29,30]。We show that inhibition of Pin1 by Juglone efficiently ameliorates both hypoxia (chronic hypoxia mouse model)- and non-hypoxia (Sugen5416 combined with chronic hypoxia rat model)-induced PAH.
总的来说,我们的数据表明,胡桃酮提供beneficial outcomes on RV hypertrophy, RV systolic pressure and RV function in both hypoxia- and non-hypoxia-induced experimental pulmonary hypertension, suggesting that targeting of Pin1 activity offers a potential therapeutic option for pulmonary hypertension.
Methods
Animals
Allin vivoprocedures were approved by local and federal animal ethics committee authorities (approval number: GI 20/10 Nr G 82/2018).
Cell culture and reagents
Human pulmonary artery smooth muscle cells (hPASMCs) were either obtained from the Universities of Giessen and Marburg Lung Center Giessen Biobank, member of DZL Platform Biobanking or purchased from Lonza (Basel, Switzerland). Murine PASMCs were isolated directly from pre-capillary pulmonary arterial vessels using iron particles, as described previously [31,32]。Human pulmonary artery endothelial cells (hPAECs) from healthy individuals were purchased from Lonza. Idiopathic pulmonary hypertension (IPAH) hPAECs were produced as described previously [33]。RV cardiac fibroblasts were isolated from adult mouse hearts, as described previously [34]。
Proliferation and apoptosis assessment
hPASMCs and hPAECs were exposed to Juglone (5-hydroxy-1,4-naphthalenedione) (sc-202675; Santa Cruz Biotechnology, CA, USA) at concentrations between 1 and 10 µM. Proliferation of hPASMCs from non-PAH individuals and patients with IPAH, and hPAECs from healthy control individuals was assayed by monitoring the incorporation of 5-bromo-2deoxyuridine (BrdU) into newly synthesised DNA (Cell Proliferation ELISA BrdU colorimetric kit; Roche, Basel, Switzerland).Apoptosis was assayed using theIn SituCell Death Detection Kit, TMR red (Roche).
Echocardiography and haemodynamic measurements
Invasive methods to measure right ventricular systolic pressure (RVSP) and systemic arterial pressure were performed on all the animals in a blinded manner. Transthoracic echocardiographic examination was performed to assess cardiac function, as described previously [35]。
Tissue preparation, histology, immunohistostaining
For histology, lungs were fixed with 10% neutral buffered formalin. The right lung was snap-frozen. The RV was separated from the left ventricle plus septum (LV+S), and the RV/(LV+S) ratio was calculated as an index of RV hypertrophy.
Statistical analysis
All data are presented as mean±sem. For comparison of two groups, parametric t-test was applied; for comparisons involving more than two groups, one-way ANOVA withpost hocNewman–Keuls multiple comparisons test was applied. Values of p<0.05 were considered statistically significant.
The details on biospecimen collections and primary cell isolations, short interfering (si)RNA transfections, luciferase reporter assays, transcription factor array, reverse transcriptase quantitative (q)PCR, Western blot analyses, immunostaining and lung morphometry are provided in thesupplementary material.
Results
Peptidyl-prolyl isomerase 1 activation in experimental and human PAH
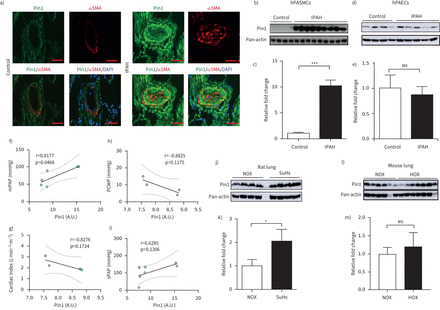
The expression of Pin1 was examined in lung specimens of patients with IPAH and non-PAH donor individuals. The immune reactivity of Pin1 was evidently augmented in pulmonary vascular compartments of IPAH patients, in comparison to non-PAH controls (figure 1a)。Co-immunostaining with α-smooth muscle actin established Pin1 localisation in the medial layer of the pulmonary arterial walls of IPAH lungs. Western blot analyses demonstrated remarkably higher Pin1 expression in IPAH hPASMCs, but not in IPAH hPAECs, as compared to their respective non-PAH control cells (figure 1b–e). Pin1 expression in IPAH hPASMCs exhibits a significant correlation with mean pulmonary arterial pressure. A weak nonsignificant correlation with pulmonary capillary wedge pressure, cardiac index and systolic pulmonary artery pressure of the IPAH patients was detected (figure 1f–i). Intriguingly, qPCR analyses of mRNA from the laser-assisted micro-dissected vessels and human lungs exhibited no significant difference ofPIN1between IPAH and non-PAH controls (supplementary figure S1a–c)。
Peptidyl-prolylcis/transisomerase, NIMA interacting 1 (Pin1) activation in experimental and human pulmonary arterial hypertension (PAH). a) Representative immunofluorescence micrograph of human lung sections from control and idiopathic PAH (IPAH) patients. Staining was undertaken for Pin1 (green) and vessel identity was visualised using α-smooth muscle actin (SMA) (red). Scale bar=50 µm. b, d) Protein expression of Pin1 in smooth muscle cells (control n=4, IPAH n=9) and endothelial cells (control n=4, IPAH n=5) isolated from pulmonary arteries of control and IPAH patients. Regulation at protein level was analysed using Western blot analysis followed by c, e) densitometric analysis. f–i) Correlation of Pin1 with clinical characteristics of IPAH patients, such as mean pulmonary arterial pressure (mPAP) (n=4, r=0.8177, p=0.0468), pulmonary capillary wedge pressure (n=6, r= −8825, p=0.1175), cardiac index (n=4, r= −0.8276, p=01724) and systolic pulmonary artery pressure (n=7, r= −0.6285, p=0.1306), respectively. j, l) Western blot analysis of Pin1 in lung homogenates exposed to Sugen5416/hypoxia (SuHx) (normoxia (NOX) n=4, SuHx n=4) and chronic hypoxia (HOX), respectively (NOX n=6, HOX n=7) followed by k, m) densitometric analysis. Pan-actin is taken as loading control. DAPI: 4′,6-diamidino-2-phenylindole; hPASMCs: human pulmonary artery smooth muscle cells; hPAECs: human pulmonary artery endothelial cells;ns:nonsignificant; A.U.: arbitrary unit. *: p<0.05, ***: p<0.001 (t-test).
An accumulation of Pin1 protein in lung homogenates of Sugen5416/hypoxia (SuHx) rats and slight, but nonsignificant enhancement of Pin1 in lungs of mice exposed to chronic hypoxia (HOX), as compared to their respective controls, was noted (figure 1j–m). Hypoxia significantly elevated Pin1 protein accumulation in cultured mouse PASMCs (supplementary figure S1d)。No significant alterations ofPin1mRNA expression have been detected in lungs of SuHx and HOX experimental models of pulmonary hypertension (supplementary figure S1e, f)。Pin1 expression is markedly elevated in pulmonary vessels of both experimental pulmonary hypertension models (supplementary figure S2a, b)。
Pin1 blockage results in suppression of vascular cell proliferation and initiation of cell apoptosisin vitro
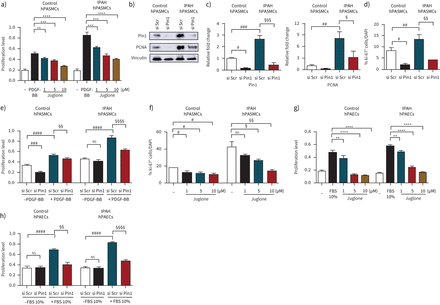
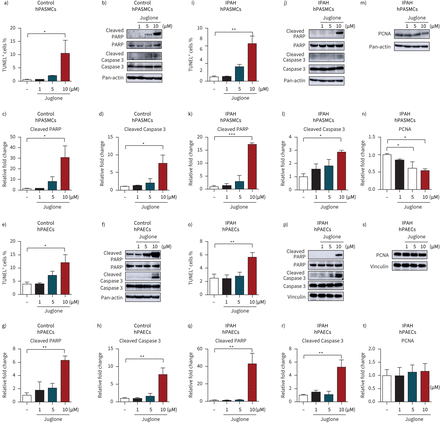
The effect of pharmacological inhibition of Pin1 by the small molecule inhibitor Juglone was first investigated on proliferation of hPASMCs. Juglone inhibited platelet-derived growth factor (PDGF)-BB-driven proliferation of both non-PAH hPASMCs and hPASMCs from IPAH patients (figure 2a)。To further support the notion that Pin1 is critically important for PASMC proliferation, an siRNA approach targeting Pin1 was employedin vitro. The expression of Pin1 was strongly reduced in Pin1-silenced hPASMCs, as compared to control transfected cells (figure 2b, c). The ablation of Pin1viasiRNA or Juglone decreased the expression of proliferating cell nuclear antigen (PCNA) and Ki-67 in hPASMCs under basal conditions (figure 2b–d, f andsupplementary figure S3a–c)。Moreover, Pin1 blockage suppressed the proliferative response of hPASMCs under basal and stimulated (PDGF-BB) conditions (figure 2e), pointing to its role in cell cycle regulation. Interestingly, the proliferation of hPAECs challenged with fetal bovine serum was also markedly inhibited by Juglone and by a Pin1-siRNA approach both in control and diseased hPAECs (figure 2g, h). Blockage of Pin1 by Juglone caused the augmentation of terminal deoxynucleotidyl transferase dUTP nick end labelling (TUNEL)-positive control and diseased hPASMCs (figure 3a, i andsupplementary figure S3d, e)。胡桃酮抑制apopto阻力sis of both control and diseased hPASMCs, as indicated by upregulated expression of pro-apoptotic active cleaved forms of Caspase 3 and poly (ADP-ribose) polymerase (PARP-1) (figure 3b–d, j–l). Pin1 inhibition led to the resistance to apoptosis of hPAECs, as determined by elevated number of TUNEL-positive control and diseased hPAECs (figure 3e, o andsupplementary figure S3f, g), increased expression of active Caspase 3 and PARP-1 apoptotic markers (figure 3f–h, p–r andsupplementary figure S3g, h)。A decrease in PCNA was detected in Juglone-treated IPAH hPASMCs (figure 3m, n), while no obvious change in IPAH hPAECs was observed (figure 3s, t). Taken together, our data suggest that Pin1 inhibition may have positive therapeutic impacts in PAH.
Peptidyl-prolylcis/transisomerase, NIMA interacting 1 (Pin1) blockage results in the suppression of vascular cell proliferationin vitro. a) Human pulmonary artery smooth muscle cells (hPASMCs) from controls and idiopathic pulmonary arterial hypertension (IPAH) patients cultured in SmGM-2 were serum-starved and treated with Juglone or dimethyl sulfoxide (DMSO) (vehicle) in the presence of platelet-derived growth factor (PDGF)-BB for 24 h. b) Representative Western blots of Pin1 and proliferating cell nuclear antigen (PCNA) expression in control and IPAH hPASMCs followed by c) densitometric analysis 24 h after Pin1 mRNA knockdown. Immunofluorescence staining for Ki-67+cells in d) Pin1-silenced (si) and f) Juglone-exposed hPASMCs. g) Human pulmonary artery endothelial cells (hPAECs) were serum-starved (0.2% fetal bovine serum (FBS) in M200) and stimulated with 10% FBS with or without Juglone for 24 h. Proliferation of Pin1-silenced e) hPASMCs and h) hPAECs of donor control and IPAH patients in presence or absence of e) PDGF-BB and h) 10% FBS determined by 5-bromo-2-deoxyuridine (BrdU) incorporation. The rate of DNA synthesis for a, e, g and h) was examined by measuring of BrdU incorporation [A370nm]. Scr: scrambled;ns:nonsignificant. Statistical analysis was performed using one-way ANOVA with Newman–Keulspost hoctest for multiple comparisons. **: p<0.01, ***: p<0.001, ****: p<0.0001versusPDGF-BB or 10% FBS treated cells;#:p<0.05,##:p<0.01,###:p<0.001,####:p<0.0001versussi scrambled or dimethyl sulfoxide (DMSO)-treated cells;§:p<0.05,§§:p<0.01,§§§:p<0.001,§§§§:p<0.0001versussi scrambled treated or IPAH cells. Data from three independent experiments are presented as mean±sem.
Peptidyl-prolylcis/transisomerase, NIMA interacting 1 (Pin1) blockage results in initiation of cell apoptosisin vitro. Terminal deoxynucleotidyl transferase dUTP nick end labelling (TUNEL) assay after 24 h treatment with increasing concentration of Juglone of a) control and i) idiopathic pulmonary arterial hypertension (IPAH) human pulmonary artery smooth muscle cells (hPASMCs), and of e) control and o) IPAH human pulmonary artery endothelial cells (hPAECs). b, j, m) Representative Western blots and c, d, k, l, n) subsequent densitometric analysis of control and IPAH hPASMCs after Juglone treatment. f, p, s) Representative Western blots and g, h, q, r, t) subsequent densitometric analysis of control and IPAH hPAECs. PARP: poly (ADP-ribose) polymerase; PCNA: proliferating cell nuclear antigen. *: p<0.05; **: p<0.01; ***: p<0.001versusdimethyl sulfoxide (DMSO)-treated control cells. Statistical analysis was performed using one-way ANOVA with Newman–Keulspost hoctest for multiple comparisons. Data from three independent experiments are presented as mean±sem.
Pin1 controls the activity of a multitude of transcription factors
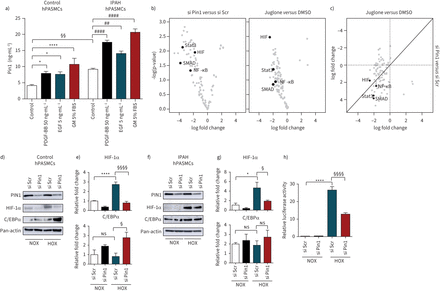
Next, we determined the effect of various pulmonary hypertension-inducing growth factors/pro-inflammatory cytokines on Pin1 expression in hPASMCs. Induction of Pin1 in hPASMCs was observed exclusively after stimulation with PDGF-BB, epidermal growth factor and growth medium, a mixture of growth factors, compared to untreated control cells (figure 4a)。Interestingly, neither members of bone morphogenetic protein family nor pro-inflammatory cytokines (tumour necrosis factor-α, interleukin-6) recognisably modified Pin1 expression, indicative of a selective mode of Pin1 regulation in hPASMCs (supplementary figure S4a–d)。As Pin1-driven isomerisation has been reported to play a significant role in the activity of various transcription factors facilitating multiple proliferation-supporting pathways [8,36], we monitored the activity of a set of 96 transcription factors from Pin1-silenced and Juglone-treated hPASMCs. The depletion of Pin1 reduced the activity of growth-promoting and increased the activity of proliferation-restraining transcription factors (figure 4b)。Several transcription factors and transcriptional co-activators implicated in pulmonary hypertension and RV dysfunction such as hypoxia-inducible factor (HIF), NF-κB, SMADs and signal transducers and activators of transcription (STATs) were dysregulated upon Pin1 inactivation either by knockdown of Pin1 or Juglone exposure (figure 4band c). A remarkable decrease of HIF-1α in control and IPAH hPASMCs and upregulation of tumour suppressor C/EBPα in control hPASMCs was detected in Pin1-silenced hypoxia-treated hPASMCs (figure 4d- g)。Hypoxia increased the hypoxia responsive element-luciferase activity that was strongly suppressed in Pin1-silenced hPASMCs (figure 4h)。
Peptidyl-prolylcis/transisomerase, NIMA interacting 1 (Pin1) controls the activity of multitude of transcription factors. a) Control and idiopathic pulmonary arterial hypertension (IPAH) human pulmonary artery smooth muscle cells (hPASMCs) after 24 h of serum starvation were subjected to platelet-derived growth factor (PDGF)-BB (50 ng·mL−1), epidermal growth factor (EGF) (5 ng·mL−1) and growth medium (GM) with 5% fetal bovine serum (FBS). Intracellular Pin1 levels were monitored by ELISA. *: p<0.05, ****: p<0.0001versuscontrol PASMCs;##:p<0.01,####:p<0.0001versusIPAH hPASMCs;§§:p<0.01 IPAH hPASMCsversuscontrol hPASMCs. Statistical analysis was performed using one-way ANOVA with Newman–Keulspost hoctest for multiple comparisons. Data from three independent experiments are presented as mean±sem. b) Pin1-silenced and Juglone-treated hPASMCs were stimulated with GM for 24 h and nuclear protein extracts were used for transcription factor activation profile array, presented as log-transformed signals in a volcano plot. c) Log-transformed scatter plot of combined transcription factor activation/inactivation in Pin1-silenced and Juglone-treated hPASMCs. Data from two independent experiments are presented. d, f) Western blots and e, g) subsequent densitometry analyses of hypoxia-inducible factor (HIF)-1α and C/EBPα transcription factors in Pin1-silenced control and IPAH hPASMCs subjected to hypoxia for 24 h. h) Hypoxia-responsive element (HRE) luciferase activity in Pin1-silenced hPASMCs after 24 h of hypoxia. Scr: scrambled;ns:nonsignificant. *: p<0.05; ****: p<0.0001 for normoxia (NOX) si Scrversushypoxia (HOX) si Scr;§:p<0.05;§§§§:p<0.0001 for HOX si ScrversusHOX si Pin1. Data from three independent experiments are presented as mean±sem.
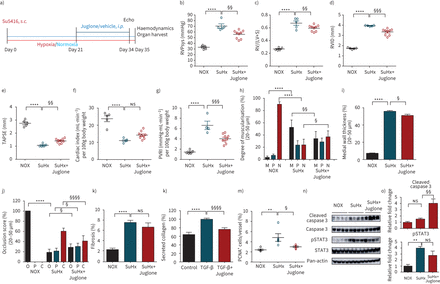
Juglone reduces pulmonary vascular remodelling and improves RV function in Sugen/hypoxia-induced PAH
The effect of Pin1 inhibition was examined in the SuHx rat model of PAH (figure 5a), closely resembling some forms of human PAH [37]。Juglone administration (1.5 mg·kg−1body weight) from day 21 to day 35 significantly reduced RVSP in comparison to placebo-treated control rats (figure 5b)。A decrease of RVSP was accompanied by diminished RV hypertrophy, as determined by the Fulton index (RV/LV+S), compared with placebo-treated control rats (figure 5c)。Administration of Juglone did not affect the systemic blood pressure (supplementary figure S4e)。The decrease in RVSP and RV hypertrophy in Juglone-treated rats was followed by diminution in RV dilation (RV internal diameter), an increase in tricuspid annular plane systolic excursion (TAPSE), nonsignificant rise in cardiac index and a severe drop in total pulmonary vascular resistance index (PVRI) (figure 5d- g)。接下来,我们决定的影响胡桃酮混乱关系atment on pulmonary vascular remodelling. Treatment of rats with Juglone reduced the number of fully muscularised pulmonary arteries and increased the number of nonmuscularised pulmonary arteries in the rat lungs, in comparison to placebo-treated control rats (figure 5handsupplementary figure S5a)。Furthermore, Pin1 inhibition resulted in a strong and significant decrease in medial wall thickness, total number of fully occluded vessels and PCNA-positive cells per vessel (figure 5i–k andsupplementary figure S5b–d), indicative of inhibition of proliferation and beneficial impact on pulmonary vessel remodelling. Next, we investigated the impact of Pin1 inhibition on right heart remodelling. Sircol assay staining of RV sections from Juglone-treated rats revealed that Pin1 inhibition provided beneficial, but nonsignificant, effects on RV fibrosis, in comparison to placebo-treated control rats (figure 5kandsupplementary figure S5e)。Transforming growth factor-β1 treatment of cardiac fibroblasts resulted in the accumulation of collagens, which was abolished by co-treatment with Juglone (figure 5l)。因此,旅游房车的纤维化损伤was associated with the reduction of collagen synthesis and secretion in cardiac fibroblasts, indicating that Pin1 is an endogenous regulator of fibrosis in right heart remodelling, secondary to pulmonary hypertension. Juglone administration strongly but nonsignificantly affected HIF-1α and C/EBPα expression (supplementary figure S5f and g)。Western blot analyses of total lung homogenates demonstrated that Pin1 blockage enhanced cleaved Caspase 3 accumulation and resulted in a clear but insignificant reduction of STAT3 activation (figure 5nand o). In summary, our data show that Pin1 inhibition improved haemodynamics, RV function and pulmonary vascular and cardiac remodelling in the SuHx model of PAH.
Juglone reduces pulmonary vascular remodelling and improves right ventricular (RV) function in Sugen/hypoxia-induced pulmonary arterial hypertension (PAH). a) Schematics of the animal treatment. Echocardiography followed by physiological measurements was performed on Juglone-treated (1.5 mg·kg−1body weight), placebo-treated Sugen5416/hypoxia (SuHx) rats and healthy rats 35 days after initiation of SuHx treatment. b) Right ventricular systolic pressure (RVPsys), as measured by right heart catheterisation, c) ratio of RV mass to mass of left ventricle plus septum (RV/(LV+S)), d) right ventricle internal diameter (RVID), e) tricuspid annular plane systolic excursion (TAPSE) (normoxia (NOX) n=5, SuHx n=5, SuHx+Juglone n=9), f) cardiac index and g) pulmonary vascular resistance index (PVRI) of the rats as measured by echocardiography. NOX n=5, SuHx n=4, SuHx+Juglone n=9.Ex vivoanalyses of lung tissues for reversal of remodelling andin vivodrug efficacy on fibrosis. h) The degree of muscularisation of small pulmonary arteries (diameter 20–50 µm) was determined by immunohistological stainings for von Willebrand factor and α-smooth muscle actin (SMA) antibodies of lung sections. M: fully muscularised; P: partially muscularised; N: nonmuscularised. i, j) Lung sections stained with Elastica van Gieson to determine medial wall thickness of vessels (%) and occlusion score (%) with a diameter of 20–50 µm. O: open; P: partial; C: closed. NOX n=5, SuHx n=5, SuHx+Juglone n=9. k) Quantitative analysis of proliferating cell nuclear antigen (PCNA)-positive cells per vessel in lungs (NOX n=4, SuHx n=4, SuHx+Juglone n=4). l, m) Effect of Juglone on RV fibrosis (NOX n=5, SuHx n=5, SuHx+Juglone n=7) and secreted collagen content in isolated cardiac fibroblasts. Data from three independent experiments are presented as mean±sem. n) Western blot analyses followed by o) densitometry of lung homogenates of representative samples from all three experimental groups. NOX n=4, SuHx n=4, SuHx+Juglone n=4.s.c.: subcutaneous;i.p.:intraperitoneal;ns:nonsignificant. **: p<0.01; ****: p<0.0001 for SuHxversusNOX;§:p<0.05;§§:p<0.01;§§§:p<0.001,§§§§:p<0.0001 for SuHx+JugloneversusSuHx. Statistical analysis was performed using one-way ANOVA with Newman–Keulspost hoctest for multiple comparisons.
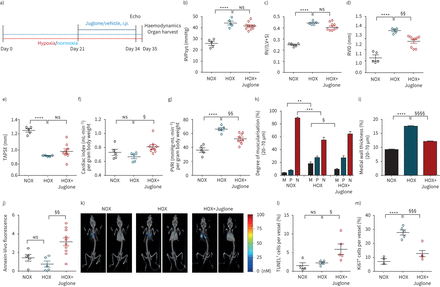
Juglone impairs the progression of pulmonary hypertension induced by chronic hypoxia
Next, the impact of Pin1 inhibition in chronic-hypoxia-induced pulmonary hypertension was examined (figure 6a)。Mice exposed to 35 days of chronic hypoxia developed pulmonary hypertension, indicated by an increase in RVSP and RV hypertrophy (figure 6b, c). Treatment with Juglone (3 mg·kg−1per day) from day 21 to day 35 resulted in a slight but nonsignificant reduction of both RVSP and Fulton index, compared to placebo-treated control mice (figure 6bc)。没有系统性血压的变化een noted after Juglone administration (supplementary figure S6e)。A slight increase in TAPSE (figure 6e), has been correlated with a substantial increase of cardiac index (figure 6f) and marked reduction in PVRI in Juglone-treated mice, as compared to placebo-treated control animals (figure 6g)。Juglone-treated mice demonstrated improved vessel remodelling, evidenced by the significant reduction of fully muscularised pulmonary arteries and medial wall thickness (figure 6h, i andsupplementary figure S6a, b)。Next, we sought to determine the impact of Juglone on apoptosis occurrence in mouse lungsin vivoandex vivo.Apoptosis was markedly and significantly increased in the lungs of Juglone-treated mice, as determined by fluorescence molecular tomography (FMT)in vivoanalyses employing Annexin-Vivo 750 probe (marks the early stages of apoptosis) (figure 6jand k). Theex vivoanalyses also revealed a significant increase in TUNEL-positive vascular cells of Juglone-treated mice (figure 6landsupplementary figure S6c) and a decrease in Ki-67+vascular cells (figure 6jandsupplementary figure S6d)。Juglone did not affect the Pin1 protein expression in Juglone-treated mice (supplementary figure S6e)。Taken together, our data demonstrate that Pin1 inhibition improves RV function and reverses pulmonary vascular remodelling in hypoxia-exposed mice.
Juglone impairs the progression of pulmonary hypertension induced by chronic hypoxia in mice. Effect of Juglone on haemodynamics and right heart function in chronic hypoxia (HOX) mice. a) Schematics of the animal treatment. Echocardiography followed by physiological measurements was carried out on Juglone-treated (3 mg·kg−1body weight), placebo-treated HOX mice and healthy mice 35 days after initiation of hypoxia treatment. b) Right ventricular systolic pressure (RVPsys) as measured by right heart catheterisation, c) ratio of right ventricle mass to mass of left ventricle plus septum (RV/(LV+S)), d) right ventricle internal diameter (RVID) and e) tricuspid annular plane systolic excursion (TAPSE). Normoxia (NOX) n=5, HOX n=5, HOX+Juglone n=10. f) Cardiac index and g) pulmonary vascular resistance index (PVRI) of the mice as measured by echocardiography. NOX n=5, HOX n=5, HOX+Juglone n=9.Ex vivoanalyses of lung tissues for reversal of remodelling. h, i) The degree of muscularisation of small pulmonary arteries (diameter 20–70 µm) was determined by immunohistology for von Willebrand factor and α-smooth muscle actin (SMA) of lung sections. M: fully muscularised, P: partially muscularised, N: nonmuscularised; lung sections stained with Elastica van Gieson were used to determine the medial wall thickness of vessels (%). j, k) Fluorescencein vivo检测到的荧光分子成像的老鼠tomography (FMT) using Annexin-Vivo 750 and representative images for all three groups. NOX n=5, HOX n=5, HOX+Juglone n=10. Apoptotic and proliferative indices determined by l) terminal deoxynucleotidyl transferase dUTP nick end labelling (TUNEL)+(氮n = 4, HOX n = 4, HOX +胡桃酮(n = 5)和m) ki - 67+cells per vessel in lungs (NOX n=5, HOX n=5, HOX+Juglone n=5).i.p.:intraperitoneal;ns:nonsignificant. **: p<0.01, ***: p<0.001, ****: p<0.0001 for HOXversusNOX;§:p<0.05,§§:p<0.01,§§§:p<0.001,§§§§:p<0.001 for HOX+JugloneversusHOX. Statistical analysis was performed using one-way ANOVA with Newman–Keulspost hoctest for multiple comparisons.
Discussion
The emergence and development of PAH encompass complex pathological mechanisms, whereas successful therapeutic treatment of the disease currently remains a major challenge. Here we show a marked elevation in the expression of Pin1 in experimental pulmonary hypertension lungs and hPASMCs of patients with PAH. Pin1 abundance in PASMCs strongly correlates with the mean pulmonary artery pressure of IPAH patients. Inhibition of Pin1 by the naphthoquinone Juglone efficiently ameliorated both hypoxia and non-hypoxia-induced PAH, evidenced by improved haemodynamics, RV function and pulmonary vascular remodelling, verifying that inhibition of disease progression associated with Juglone-mediated effects on Pin1. These data are in correlation within vitroresults, in which Pin1 blockage diminished proliferation and declined the resistance to apoptosis of hPASMCs and hPAECs. Pin1 is an enzyme that catalyses thecis/transconversion of its substrate upon binding to the pSer/Thr-Pro consensus motifs within the target [10,38,39]。Pin1-driven isomerisation of multiple substrates results in various biological outcomes [22,40], providing an alternative path of control of abundant signalling cascades under different cellular conditions [14,41]。Pin1 controls the activity of a range of transcription factors and could provide a pivotal switch in disease pathogenesis by regulating the gene expression of its transcriptional substrates [42]。Pin1 contributes to cell cycle control of various diseases [22,40,43,44], partially by regulating the nucleo-cytoplasmic shuttling and activity of transcription factors, which control cell proliferation and inflammatory response. For example, Pin1-mediated nuclear shuttling and isomerisation of β-catenin leads to upregulation of its target genes, Cyclin D1 and c-Myc [45]。Pin1 promoted nuclear accumulation of NF-κΒ subunit RelA,viainhibition its binding to ΙκΒα, enhances cell growth and inflammatory cytokine production [46]。Pin1 could also sequester the cytoplasmic shuttling of a tumour suppressor of forkhead family of transcription factor 4 (FOXO4), resulting in a decrease of its transcriptional activity toward its target genes, such as p27kip1 [47]。Pin1 can regulate the stability of plethora of transcription factors, key regulators of PAH emergence and progression, as FOXOM [48,49]; peroxisome proliferator-activated receptor-γ [50,51], HIF [52,53], oestrogen receptor-α [54,55] and transcriptional co-activators, such as Notch [56,57]。Moreover, Pin1 has been reported to increase the stability and transcriptional activity of bromodomain-containing protein 4 (BRD4), one of the critical epigenetic drivers for PAH [58,59]。In our study, transcription factor profiling arrays revealed that Pin1 efficiently controls the activity of key transcription factors, implicated in pulmonary hypertension emergence and development, suggesting that Pin1 is an important endogenous integrator in the context of PAH. Specifically, we show that siRNA-mediated or Juglone-treated depletion of Pin1 resulted in a strong decrease in transcriptional activity of crucial transcription factors, which have been implicated previously in cell proliferation and migration, chronic inflammation and tissue remodelling (STAT3, HIF, NF-κB and SMADs) [60–62]。This agrees with observations that Pin1 blockage resulted in a marked decrease in proliferative responses of hPASMCs and hPAECs, indicating that endogenous Pin1 contributes to the pseudomalignant phenotype of the disease. Anti-apoptotic resistance of vascular cells is another hallmark of PAH [3], and Pin1 has been implicated in cell death in various pathological conditions [63,64]。Importantly, a strong and significant initiation of apoptosis and successive inhibition of vascular cell proliferation distinguished upon Pin1 deletion was tightly correlated with a substantial initiation of cell death in lungs of chronic-hypoxia-induced pulmonary hypertension. Knockdown of Pin1 in pulmonary vascular cells resulted in inhibition of proliferative and activation of pro-apoptotic responses, implicating the endogenous Pin1 for pulmonary hypertension development. Myocardial apoptosis has been associated with RV dysfunction and fibrosis [65]; thus, apoptosis may provide a detrimental impact on RV function. In our study, apoptosis initiation was exclusively presented in lung vascular cells, and more specifically in smooth muscle cells, while FMT did not reveal any apoptotic signal in the heart. Pin1 regulates the intensity and duration of cardiac hypertrophic response [66]。The inhibition of Pin1 alleviated cardiac damage and fibrosis in isoprenaline- and diabetes-induced myocardial fibrosis in rats [67,68], verifying our data that Pin1 contributes to the development of cardiac remodelling in SuHx-administered rats. Furthermore, recently it was reported that Pin1 blockage reversed the PAH phenotype in PAH microvascular endothelial cellsin vitroand in PAH ratsin vivo[69]。Interestingly, our detailed expression analyses of Pin1 indicated inconsistency between mRNA and protein both in experimental and clinical PAH, suggesting that Pin1 protein might be stabilised in the course of the disease. Plk-1 (whose expression was recently shown to be significantly increased in distal pulmonary arteries and isolated PASMCs from PAH patients; Sébastien Bonnet, Pulmonary Hypertension and Vascular Biology Research Group, Institut Universitaire de Cardiologie et de Pneumologie de Québec, Université Laval, Department of Medicine, Québec, Canada; personal communication) has been reported to stabilise Pin1 protein, which might explain the disparity in mRNA and protein levels of Pin1 in PAH [23]。
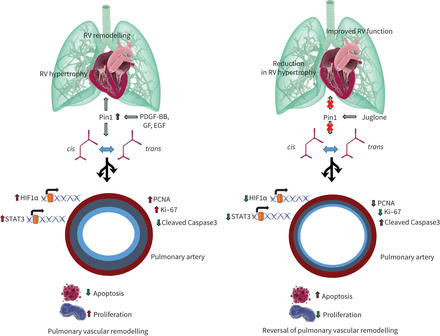
Juglone, along with another naphthoquinone, Plumbagin, is intensively studied in cancerin vivo[70–73]。Plumbagin-inhibited PAH-hPASMC proliferation and resistance to apoptosis were associated with a decrease in pulmonary artery remodelling, mean pulmonary artery pressure and RV hypertrophy in experimental PAH rat modelsviathe STAT3/NFAT axis [74]。The deletion of Pin1 in hPASMCs in our experiments also resulted in a strong and remarkable decline of both STAT3 and STAT5 activity, suggesting a common route of regulation for both naphthoquinones. One of the primary limitations of this study is that the control and PAH-hPAECs were obtained from different sources. The slight variations in the isolations and culturing procedures of these primary cells may impact the functional activity of these cells in response to Juglone. Many of naphthoquinones including Juglone are characterised by pro-oxidant properties. In this regard, Juglone acts not only as a redox-cycling agent, but also as producer of reactive oxygen species [75]。In addition to its pro-oxidant properties, Juglone also possess both the cytotoxic and the genotoxic properties [76], thus the damaging impact of Juglone on health cells cannot be completely ruled out. Pin1 serves as a linkage between various signalling mechanisms at multiple levels. A proposed Pin1 mechanism in pulmonary hypertension has been illustrated infigure 7.
A proposed signalling mechanism of peptidyl-prolylcis/trans异构酶,尼玛互动1 (Pin1)在肺hypertension. Aberrant growth factor signalling induces Pin1 leading to isomerisation of various target proteins and regulation of transcription factors, resulting in proliferation and survival of pulmonary vascular cells. Amplification of pro-proliferative and anti-apoptotic pathways by Pin1 leads to pulmonary vascular remodelling and right ventricular (RV) failure and Juglone, by inhibiting Pin1 response, could provide a potential prevention strategy for pulmonary hypertension. PDGF: platelet-derived growth factor; GF: growth factors; EGF: epidermal growth factor; HIF: hypoxia-inducible factor; STAT: signal transducers and activators of transcription; PCNA: proliferating cell nuclear antigen.
Our data underscore the importance of Pin1 for controlling the activity of presently uninvestigated transcription factors in PAH. A deeper understanding of the molecular mechanisms driving Pin1 regulation and the molecular/cellular circuits driven by Pin1 in diseased vascular cells is essential for developing tailored therapeutic concepts focusing on Pin1 inhibition.
Supplementary material
Supplementary Material
Please note:supplementary material is not edited by the Editorial Office, and is uploaded as it has been supplied by the author.
Supplementary materialERJ-01698-2021.Supplement
Shareable PDF
Supplementary Material
This one-page PDF can be shared freely online.
Shareable PDFERJ-01698-2021.Shareable
Acknowledgements
The authors would like to thank Carina Lepper, Christina Vroom, Ewa Bieniek and Sophia Hattensohl (Excellence Cluster Cardio Pulmonary Institute (CPI), Giessen, Germany) for their technical assistance.
Footnotes
Conflict of interest: W. Seeger reports personal fees from Actelion, Bayer AG, Novartis, Vectura, Medspray and United Therapeutics, outside the submitted work. All other authors disclose no potential conflicts of interest.
Support statement: The authors received funding from the German Research Foundation (DFG) by the CRC1213 (Collaborative Research Center 1213) projects A08, B04 and CP02. Funding information for this article has been deposited with theCrossref Funder Registry.
- ReceivedFebruary 19, 2020.
- AcceptedDecember 29, 2021.
- Copyright ©The authors 2022.
This version is distributed under the terms of the Creative Commons Attribution Non-Commercial Licence 4.0. For commercial reproduction rights and permissions contactpermissions{at}ersnet.org