Abstract
BackgroundThe relationship between obstructive sleep apnoea (OSA) and cognitive decline remains controversial, especially in the elderly population. We used data from the HypnoLaus study to assess associations between OSA and longitudinal cognitive changes in a sample of community-dwelling elderly individuals.
MethodsWe studied associations between polysomnographic OSA parameters (of breathing/hypoxaemia and sleep fragmentation) and cognitive changes over a 5-year period, after adjustment for potential confounders. The primary outcome was the annual change in cognitive scores. The moderating effects of age, sex and apolipoprotein E4 (ApoE4) status were also examined.
Results358 elderly individuals without dementia were included (mean±sdage 71.0±4.2 years; 42.5% males). A lower mean peripheral oxygen saturation (SpO2) during sleep was associated with a steeper decline in Mini-Mental State Examination (B= −0.12, p=0.004), Stroop test condition 1 (B=0.53, p=0.002) and Free and Cued Selective Reminding Test delayed free recall (B= −0.05, p=0.008). A longer time spent asleep withSpO2<90% was associated with a steeper decline in Stroop test condition 1 (B=0.47, p=0.006). Moderation analysis showed that apnoea–hypopnoea index and oxygen desaturation index were associated with a steeper decline in global cognitive function, processing speed and executive function only in older participants, men and ApoE4 carriers.
Conclusions我们的研究结果提供的证据贡献啊f OSA and nocturnal hypoxaemia to cognitive decline in the elderly population.
Abstract
In a sample of elderly people from the general population, obstructive sleep apnoea parameters, particularly those of nocturnal hypoxaemia, predicted steeper cognitive decline over 5 yearshttps://bit.ly/3RsvsX3
Introduction
Obstructive sleep apnoea (OSA) is a chronic condition characterised by recurrent upper airway obstructions during sleep. OSA is common, especially in the elderly, with an estimated prevalence of at least 30% [1]。Increasing evidence indicates that OSA has a detrimental effect on brain health [2], yet its role in cognitive decline remains controversial in the elderly population [3–5]。Seminal prospective studies conducted in elderly individuals have suggested that OSA is a risk factor for the development of mild cognitive impairment and dementia [6–8]。However, the few studies that have examined the association between objectively measured OSA and longitudinal changes in cognitive performance (as measured by cognitive tests) have reported mixed results [9–12]。These studies have shown no association [9,10] or only modest associations [11,12] between OSA and cognitive decline in the elderly population.
As cognitive decline in old age can result from many different factors, assessing the specific contribution of OSA appears to be a difficult task [4,5]。The question remains whether the impact of OSA on cognitive decline varies according to the presence of non-modifiable risk factors for dementia, such as older age, female sex or the presence of the apolipoprotein E4 (ApoE4) allele [13]。The ApoE4 allele is the strongest genetic risk factor for sporadic Alzheimer's disease [14] and is associated with poorer performance in various cognitive domains, even in elderly people without dementia [15]。
A systematic review observed weaker associations between OSA and cognitive impairment in studies conducted in the elderly than in studies conducted in young and middle-aged adults [16]。此外,两个队列研究associ报道ations between OSA and poorer cognitive function in middle-aged participants, but not in the elderly [17,18]。This has led to the hypothesis that ageing is associated with a decrease in the strength of the association between OSA and cognitive impairment, potentially because the effect of OSA is blurred by other factors that impact cognition [13]。The two cohort studies also suggested a stronger relationship between OSA and cognitive impairment in women than in men [17,18]。Furthermore, an epidemiological study found that women (but not men) with OSA were more likely to develop dementia over a 5-year period than age- and sex-matched controls without OSA [19]。Taken together, these studies suggest that women with OSA may have a higher risk of cognitive decline than men [17–19]。Finally, most previous evidence, although based on cross-sectional studies, suggests a stronger association between OSA and cognitive impairment in ApoE4 carriers [20–22]。However, only a part of these studies empirically examined the moderating effect of age, sex or ApoE4 using moderation analysis [17,21,22], whereas the remaining studies performed a stratified analysis without assessing the interaction terms [18–20]。
The present study aimed to determine associations between OSA and cognitive decline over 5 years in a sample of community-dwelling elderly people. We also performed moderation analysis to examine the effect of age, sex and ApoE4 on the association between OSA and cognitive changes. We expected OSA to have weak associations with cognitive decline in the whole sample, but that these associations would be stronger in younger participants, women and ApoE4 carriers.
Methods
Study population
Data came from participants of the CoLaus|PsyCoLaus study, a prospective cohort of middle-aged and older adults randomly selected from the residents of Lausanne, Switzerland [23,24]。Polysomnography (PSG) was performed during the first follow-up. Cognitive assessment was performed in participants aged ≥65 years at the first and second follow-up. The present study included participants, aged ≥65 years, who underwent PSG and a cognitive assessment at the first follow-up (baseline of the present study), followed by a second cognitive assessment 5 years later (follow-up of the present study). We excluded participants without ApoE genotyping, with dementia (defined as a Clinical Dementia Rating score ≥1 [25]) and with central sleep apnoea (supplementary figure S1) [26]。The CoLaus|PsyCoLaus and HypnoLaus studies were approved by the ethics committee of the Vaud Canton and all participants provided written informed consent.
OSA parameters
Home-based PSG recordings were performed as part of the HypnoLaus study (seesupplementary materialfor more details) [1]。PSG was performed a mean±sd0.97±0.94 years after the first cognitive assessment. Respiratory events were scored according to the 2012 American Academy of Sleep Medicine criteria [27]: apnoea was defined as a ≥90% decrease in airflow from baseline lasting ≥10 s and hypopnoea was defined as a ≥30% decrease in airflow lasting ≥10 s with either an arousal or a ≥3% oxygen desaturation. We selected six OSA parameters of breathing/hypoxaemia and sleep fragmentation. These parameters were analysed as continuous and categorical variables: apnoea–hypopnoea index (AHI; <15versus≥15 events·h−1), oxygen desaturation index ≥3% (ODI; <15versus≥15 events·h−1), mean peripheral oxygen saturation during sleep (meanSpO2; ≤92.5%=lower quartileversus>92.5%), sleep time withSpO2<90% (TST90; <4.5%versus≥4.5%=upper quartile), sleep efficiency (≤72%=lower quartileversus>72%) and arousal index (<32versus≥32 events·h−1=上四分位数). In addition, we also studied hypoxic burden [28] as a continuous and categorical variable (<42versus≥42%min·h−1=上四分位数); 18 participants had missing data for this variable due to technical failure and/or insufficient quality of the pulse oximeter signal.
Cognitive assessment
Cognitive assessment included the Mini-Mental State Examination (MMSE; global cognitive function) [29], Stroop test Victoria version (Stroop test; processing speed and executive function) [30], verbal fluency tasks (executive control and verbal ability) [31], Free and Cued Selective Reminding Test (FCSRT; episodic verbal memory) [32,33], 40-DO naming test (language) [34] and constructional praxis task from the Consortium to Establish a Registry for Alzheimer's Disease neuropsychological battery (visuospatial function) [35]。The primary outcome was the annual change in cognitive scores (=(scorefollow-up–scorebaseline)/years of follow-up). Negative annual changes corresponded to a cognitive decline for all tests except the Stroop test, for which positive values corresponded to a decline. The secondary outcome was the incidence of significant cognitive decline, defined as a decrease in performance of >1.0sdabove the mean annual change (table 1andsupplementary table S1).
Cognitive assessment
Potential confounders and moderators
Potential confounders and moderators were recorded at the baseline of the present study. Education level was dichotomised into
Statistical analysis
For descriptive analysis, we categorised the sample into subgroups with an AHI <15versus≥15 events·h−1. Normal distribution of continuous variables was tested using Q–Q plots. Between-group differences in baseline characteristics were analysed using the unpaired t-test, Mann–Whitney U-test, Chi-squared test or Fisher's exact test, as appropriate. Associations between OSA parameters and annual change in cognitive scores were assessed using linear regression. Assumptions of the linear regression were tested (no violation was observed). Findings were expressed as unstandardised β-coefficient (B)和95%置信区间。分析与我ncidence of significant cognitive decline as the outcome variable were conducted using logistic regression. Goodness-of-fit of the model was assessed through the Hosmer–Lemeshow test (no evidence of poor fit was observed). Findings were expressed as odds ratio and 95% confidence interval. Three models were constructed to further explore the impact of confounders. Model 1 was adjusted for age, sex, education and ApoE4. Model 2 was additionally adjusted for BMI, diabetes, hypertension, smoking, alcohol consumption and psychotropic drugs. Model 3 was additionally adjusted for depression, ESS, CPAP and COPD. We used Hayes’ SPSS Process macro version 4.1 [37] to explore the moderating effects of age, sex and ApoE4 on the association between OSA parameters and cognitive changes, with adjustment for all of the aforementioned confounders. For the assessment of age as a potential moderator, we used the Johnson–Neyman technique to determine the age range for which the moderator effect was significant [37]。Findings were expressed as interaction term (OSA parameter×moderator) and conditional effects of the OSA parameter at the moderator values. The sensitivity analyses tested: 1) additional AHI thresholds (≥20, ≥25 and ≥30 events·h−1), 2) hypoxic burden as a predictor (after excluding the 18 participants missing these data) and 3) the impact of excluding participants treated with CPAP (n=8). A description of how missing data were handled is provided in thesupplementary material. The level of significance was set at two-sided p<0.05 for descriptive analysis and lowered to p<0.01 for regression and moderation analyses.
Results
Baseline characteristics
The final sample consisted of 358 community-dwelling elderly individuals with a mean±sdage of 71.0±4.2 years, of whom 152 (42.5%) were male (table 2). The average follow-up time between the two cognitive assessments was 5.2±0.5 years. At baseline, participants with AHI ≥15 events·h−1were more likely to be men, had higher BMI, had higher prevalence of diabetes and hypertension, were more likely to be smokers, had higher alcohol consumption, and had higher ESS score. All PSG measures of interest were significantly different between groups. The AHI ≥15 events·h−1group was more likely to have lower performances on the MMSE, Stroop test conditions 2 and 3, and FCSRT delayed free recall. Baseline characteristics of included participantsversusindividuals who were excluded because they did not complete the second cognitive assessment are shown insupplementary table S2. The excluded individuals had higher prevalence of diabetes, higher use of psychotropic drugs, lower prevalence of depression, higher AHI and ODI, lower sleep efficiency, higher arousal index, and lower performance on some cognitive tests.
Baseline characteristics of the sample
Annual change in cognitive scores
Table 3shows significant associations between OSA parameters and annual change in cognitive scores (complete results are presented insupplementary table S3). Participants with meanSpO2≤92.5% had a steeper decline in MMSE (B= −0.12, p=0.004, according to Model 3) and Stroop test condition 1 (B=0.53, p=0.002, according to Model 3). Lower meanSpO2(as a continuous variable) was associated with decreased performance in FCSRT delayed free recall (Bfor 1% decrease= −0.05, p=0.008, according to Model 3). Participants with TST90 ≥4.5% also had a greater annual decline in Stroop test condition 1 (B=0.47, p=0.006, according to Model 3).
Associations between obstructive sleep apnoea (OSA) parameters and annual change in cognitive scores
Incidence of significant cognitive decline
Significant associations between OSA parameters and incidence of significant cognitive decline are presented intable 4(complete results are reported insupplementary table S4). MeanSpO2≤92.5% was associated with increased odds of significant decline in MMSE (OR 3.27, p=0.001, according to Model 3; corresponding to a decline of ≥2.0 points during the follow-up) and in Stroop test condition 1 (OR 4.64, p=0.007, according to Model 3; corresponding to an increase of ≥7.3 s to complete the task during follow-up). In Model 1, lower meanSpO2(as a continuous variable) was associated with incidence of significant decline in FCSRT delayed free recall (OR for 1% decrease 1.21, p=0.007; corresponding to a decline of ≥3.2 points during follow-up); a trend towards significance was observed in the other models (OR 1.22, p=0.010, according to Model 2 and OR 1.22, p=0.011, according to Model 3).
Associations between obstructive sleep apnoea (OSA) parameters and incidence of significant cognitive decline
Moderation analysis
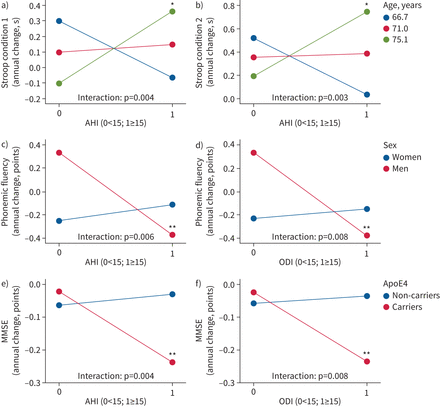
Significant moderating effects are presented intable 5andfigure 1(baseline characteristics of the sample according to age, sex and ApoE4 status are shown insupplementary tables S5–S7). AHI ≥15 events·h−1was associated with a steeper decline in Stroop test condition 1 and condition 2 only in older participants (p<0.05 for participants aged >75 years and p<0.01 for those aged >78 years). AHI ≥15 events·h−1and ODI ≥15 events·h−1were associated with a more pronounced decline in phonemic fluency only in men (B= −0.70, p=0.003 andB= −0.71, p=0.002, respectively). AHI ≥15 events·h−1and ODI ≥15 events·h−1were associated with a greater decline in MMSE only in ApoE4 carriers (B= −0.21, p=0.007 andB= −0.21, p=0.009, respectively). No significant moderating effect was observed for the associations between OSA parameters and significant cognitive decline.
Characteristics moderating the association between obstructive sleep apnoea (OSA) parameters and cognitive changes
Conditional effects of obstructive sleep apnoea (OSA) parameters at moderator values. Data were analysed by Hayes' SPSS Process macro version 4.1 [37] for moderation analysis using annual change in cognitive score as dependent variable, OSA parameter as independent variable and a, b) age, c, d) sex or e, f) apolipoprotein E4 (ApoE4) status as moderator. Asterisks indicate significant difference between apnoea–hypopnoea index (AHI; events·h−1) or oxygen desaturation index (ODI; events·h−1) groups: *: p<0.05; **; p<0.01. Models were corrected for age (continuous), sex (male, female), education (≥high school,
Sensitivity analyses
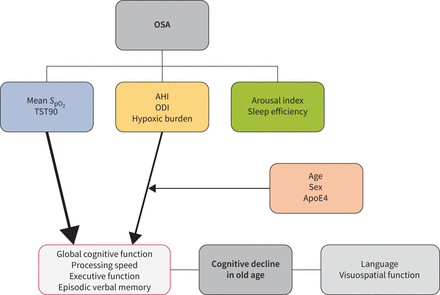
Participants with AHI ≥20 events·h−1were more likely to show significant decline in phonemic fluency (OR 2.99, p=0.003, according to Model 3; corresponding to a decline of ≥7.6 points during follow-up) (table 4, andsupplementary tables S8 and S9). Participants with hypoxic burden ≥42%min·h−1显示趋势likeliho更高od of significant decline in phonemic fluency (OR 2.26, p=0.033, according to Model 3) (supplementary tables S10 and S11). The exclusion of participants treated with CPAP had no impact on the results (data not shown).Figure 2provides an overview of the main results of the study.
Overview of the results. The thickness of the arrows indicates the strength of the associations. Mean peripheral oxygen saturation during sleep (meanSpO2) and sleep time withSpO2<90% (TST90) were the parameters most consistently associated with cognitive decline. Apnoea–hypopnoea index (AHI), oxygen desaturation index (ODI) and hypoxic burden were less consistently associated with cognitive decline, and their associations were partly moderated by age, sex and apolipoprotein E4 (ApoE4) status. OSA: obstructive sleep apnoea.
Discussion
In this cohort of community-dwelling elderly individuals, AHI and indices of nocturnal hypoxaemia (but not those of sleep fragmentation) were associated with steeper cognitive decline over 5 years. This decline was observed in global cognitive function, processing speed, executive function and episodic verbal memory, but not in language or visuospatial function. MeanSpO2and TST90 were the parameters most consistently related to changes in cognitive functioning. AHI and ODI were specifically associated with steeper cognitive decline in older participants, men and ApoE4 carriers. This study provides evidence of the contribution of OSA to cognitive decline in the elderly population. It also highlights the detrimental role of hypoxaemia and the presence of demographic and genetic moderating factors.
Two previous cohort studies have examined the relationship between PSG measures of OSA and cognitive changes in the elderly [9,12]。The Osteoporotic Fractures in Men Study found an association between TST90 and 3-year decline in Modified MMSE in 2636 men aged 76 years [12]。However, in the Atherosclerotic Risk in Communities study, which included 966 participants aged 61 years, there was no association between OSA parameters and 15-year changes in the three cognitive tests examined [9]。Two other studies have used non-PSG respiratory devices to assess the association between OSA and cognitive decline [10,11]。In an analysis of the Proof-Synapse Cohort, in which 559 participants (aged 67 years) were followed over 8 years, AHI ≥30 events·h−1was associated with greater decline in the attentional domain [11]。Furthermore, AHI (as a continuous variable) and parameters of hypoxaemia were associated with changes in attention and executive function [11]。These results contrast with those of the Hispanic Community Health Study/Study of Latinos (HCHS/SOL), which showed no association between the presence of sleep disordered breathing (defined as a respiratory event index ≥15 events·h−1) and 7-year cognitive decline in a sample of 5247 participants with a mean age of 63 years [10]。Similarities with our study include the relatively weak association between AHI and cognitive changes [11], the role of measures of hypoxaemia [11,12], and the decline observed in global cognitive function and executive function [11,12]。Discrepancies may be due to differences in demographics (age and sex of the samples), measures of OSA (PSGversusnon-PSG devices), follow-up time and cognitive tests.
The moderation analysis was a novel aspect of this study. AHI and ODI were associated with a steeper decline in some cognitive tests only in older participants, men and ApoE4 carriers. Results concerning the effect of ApoE4 agreed with our hypothesis, while those concerning the effect of age and sex did not. Our results of an increased risk of OSA-related cognitive decline in ApoE4 carriers are in line with most previous cross-sectional studies [20–22]。However, further longitudinal investigations are needed to corroborate this hypothesis.
We expected a stronger association between OSA and cognitive decline in younger than older participants. This speculation was based on a systematic review [16] and two cross-sectional cohort studies using data from the HCHS/SOL [17] and the Canadian Longitudinal Study of Aging (CLSA) [18]。Age-stratified analysis of these cohort studies showed that associations between AHI (measured by respiratory polygraphy) [17] or OSA risk (defined by the combination of STOP score and whole-body fat percentage) [18] and poorer cognitive performance were more evident in middle-aged than in older women. Our results, combined with previous findings, may indicate a non-linear relationship between OSA and cognitive impairment in ageing, with more pronounced associations visible before 60 and after 75 years of age; however, this hypothesis requires further investigation.
As already mentioned, findings from the HCHS/SOL and the CSLA suggest a stronger relationship between OSA and cognitive impairment in women than in men [17,18]。Furthermore, in an epidemiological study based on data from Taiwan's National Health Insurance Research Database, only women withversuswithout OSA were more likely to develop dementia over a 5-year period [19]。However, these studies included women in the perimenopausal period (45–54 years) [17–19] and associations between OSA and cognitive impairment were mostly evident during this age span [17,18]。This suggests that perimenopause may increase women's vulnerability to the detrimental effects of OSA on brain health [13]。The fact that our sample did not include women in the perimenopausal period may partly explain our results. Moreover, the prevalence of OSA in women increases mainly during the perimenopause, whereas in men OSA often appears at a younger age. Therefore, the negative effects of OSA on cognition in men may accumulate over a greater number of years than in women.
阻塞性睡眠呼吸暂停综合症参数与更大的下降n global cognitive function (MMSE), processing speed (Stroop test condition 1 and condition 2), executive function (phonemic fluency) and episodic verbal memory (FCSRT delayed free recall), whereas no association was observed for language or visuospatial function. This pattern of cognitive changes is consistent with previous literature [3,16] and appears to be similar to that seen in frontal-subcortical syndrome, which is mainly characterised by dysexecutive symptoms and decreased psychomotor speed [38]。One of the main causes of frontal-subcortical syndrome are microvascular lesions in the white matter and the deep grey nuclei, which can lead vascular dementia [39]。This suggests that OSA may promote microvascular pathology that ultimately results in cognitive impairment in the elderly, as previously postulated [40]。In support of this hypothesis, it has been shown that OSA and nocturnal hypoxaemia are associated with frontal white matter lesions [41,42] and morphometric changes in the deep grey nuclei [43,44]。In addition, it has recently been reported that neurofilament light chain (a marker of white matter integrity) increases in plasma of patients with severe OSA as a function of TST90 after CPAP withdrawal [45]。Decline in episodic verbal memory only concerned the delayed free recall task, but not the delayed total recall task. This also appears consistent with frontal-subcortical syndrome, as a poor free recall performance associated with a normal total recall suggests reduced memory retrieval (which is dependent on the frontal lobe) rather than reduced memory encoding/storage (which is dependent on the medial temporal lobe) [46]。
From a clinical perspective, our results suggest that individuals with OSA who have more severe hypoxaemia are at increased risk of developing cognitive impairment. Intriguingly, meanSpO2and TST90 were more consistently related to cognitive decline than intermittent hypoxaemia parameters. This may suggest that the brain is more vulnerable to lower basal oxygen levels during sleep than to episodes of intermittent hypoxaemia; however, this hypothesis requires further investigation. Older age, male sex and ApoE4 also appear to be vulnerability factors for cognitive decline in OSA. These insights may be useful in identifying “at-risk” OSA patients who can be selected for interventional studies. The implications are important because cognitive impairment and dementia are often irreversible conditions for which management of modifiable risk factors is of paramount importance.
To date, it remains unclear whether CPAP treatment prevents or delays cognitive decline in the elderly. Small single-centre trials have shown improvements in cognitive functioning during CPAP treatment [47,48]。In contrast, larger multicentre trials have shown no effect [49,50] or only slight improvement in cognition [51,52]。Nevertheless, it should be mentioned that the benefit of CPAP is expected to increase with good adherence and that low adherence has been reported in some of these trials [50,52]。Moreover, these trials mostly included highly educated participants with high baseline cognitive scores, which could have led to a ceiling effect [49–52]。
The strengths of our study were the longitudinal design, PSG measures of OSA, cognitive assessment using an extensive battery of tests, availability of information about many potential confounders and moderating analysis. Limitations were the assessment of sleep on a single night only because studies have reported night-to-night variations in the assessment of OSA [53]。然而,multi-night PSG录音是罕见的both clinical and research settings, and were not available in our cohort. We had to exclude participants who did not complete the second cognitive assessment. This could have led to selection bias because, as mentioned earlier, the included compared with excluded sample had some more favourable characteristics at baseline. Finally, the results should be interpreted with caution because we examined a 5-year time window only, and some relevant associations between OSA and cognitive decline might lie outside this window.
In conclusion, OSA parameters (particularly those measuring nocturnal hypoxaemia) were independent predictors of 5-year cognitive decline in our sample of community-dwelling elderly individuals. Older age, male sex and ApoE4 emerged as vulnerability factors exacerbating this association. This study provides new insights that may be useful for identifying OSA patients at increased risk of cognitive decline and for designing new interventional studies.
Supplementary material
Supplementary Material
Please note:supplementary material is not edited by the Editorial Office, and is uploaded as it has been supplied by the author.
Supplementary materialERJ-01621-2022.SUPPLEMENT
Shareable PDF
Supplementary Material
This one-page PDF can be shared freely online.
Shareable PDFERJ-01621-2022.Shareable
Acknowledgements
We thank all the participants in the CoLaus|PsyCoLaus and HypnoLaus studies. English language editing assistance was provided by Nicola Ryan (independent medical writer, Auckland, New Zealand) funded by Lausanne University Hospital and University of Lausanne, Lausanne, Switzerland.
Footnotes
Conflicts of interest: R. Heinzer is member of the medical advisory board of Dreem and Nightbalance (Philips), and received speaker's fees or honorarium from ResMed, Philips, Jazz and Inspire. The remaining authors have no potential conflicts of interest to disclose.
This article has an editorial commentary:https://doi.org/10.1183/13993003.00300-2023
Support statement: The CoLaus|PsyCoLaus and HypnoLaus studies were supported by GlaxoSmithKline, Faculty of Biology and Medicine of the University of Lausanne, Swiss National Science Foundation (grants 3200B0-105993, 3200B0-118308, 33CSCO-122661, 33CS30-139468, 33CS30-148401 and 33CS30-177535), Leenaards Foundation, and Vaud Pulmonary League (Ligue Pulmonaire Vaudoise). The present study was supported by the Swiss National Science Foundation (Postdoc.Mobility grant to N.A. Marchi). Funding information for this article has been deposited with theCrossref Funder Registry.
- ReceivedAugust 18, 2022.
- AcceptedJanuary 29, 2023.
- Copyright ©The authors 2023.
This version is distributed under the terms of the Creative Commons Attribution Non-Commercial Licence 4.0. For commercial reproduction rights and permissions contactpermissions{at}ersnet.org