抽象的
Repeated measures of eosinophil counts increased the proportion of eosinophilic patientshttp://ow.ly/LhnV30hlR5j
To the Editor:
血液嗜酸性粒细胞作为慢性阻塞性肺病(COPD)中的生物标志物存在相当大的争议。最近的文献表明它用于评估对吸入皮质类固醇的反应(IC)的反应[1,2] and in predicting exacerbations [3,4]。其他研究完全或部分反驳这些关联[5–7]。We suggest that what underlies this confusion is the failure to understand the temporal variation in the biomarker, and the inherent imprecision in the collection of associated end-point data.
血液嗜酸性粒细胞受到许多因素的影响,包括昼夜,季节性和激素变化,四十年来已知[8,9]。在COPD中,疾病和治疗的波动进一步增加了这种变异性[10]。Thus, the reliance on a single estimation of eosinophil count at baseline of a randomised control trial would be highly unlikely to reflect accurately the overall pattern of blood eosinophilia, and therefore the relationship with exacerbation risk or treatment response. Recently, a minority of patients were shown to have persistent eosinophilia, with the majority either non- or episodically eosinophilic [11]。作为R.abeet al.[12]指出,主要问题是需要多少估计,并且一个人在哪里画出线?
We have retrospectively collected “real-world” data on the prevalence and variability of blood eosinophilia in COPD patients, both with and without a co-existing diagnosis of asthma, as part of an on-going audit to assess compliance with our local guidelines. We manually examined primary care electronic records for patients on the COPD register of general practices in the Hull and East Riding area of the UK. The last five available eosinophil counts were collected. The most recent eosinophil count was termed E1, the penultimate E2, and so on, up to a maximum of five eosinophil counts (E5). A COPD exacerbation was defined as requiring prescription of short-term oral steroids for COPD within 12 months of data collection.
Any absolute eosinophil count (E1–E5) of ≥0.30×109每升细胞被选为我们的嗜酸性粒细胞的截止点,以镜像当地指南[13]。However, we also assessed ≥0.34×109[4] and ≥0.45×109[3], as used in previous reports. The proportion of eosinophilia was plotted against E1–E5. Continuous variables were described as median (interquartile range). Quasi-Poisson regression [14] adjusted for age, gender, predicted forced expiratory volume in 1 s (FEV1), the Medical Research Council (MRC) dyspnoea scale, smoking status and asthma co-diagnosis was employed, and adjusted rate ratios and 95% confidence intervals are reported. Missing data was handled with 40 multiple imputation datasets [15],使用预测平均匹配的连续变量,对二分变量的逻辑回归和MRC秤的有序Logit回归。根据Rubin的规则汇集了结果[16,17]。R版本3.4.1软件用于统计分析。P值<0.05 / 5(Bonferroni校正)被认为是统计学意义的。
There were 611 (320 male) patients in our cohort, from four general practices. 253 patients had a co-diagnosis of asthma. The median age was 68 (60–76) years and predicted FEV1是62%(46-75%)。非吸烟者的队列(611分中为362人),而40.6%(611分中,248人)是目前吸烟者。
我们的队列中有95%(581人)我们的队列至少有一个预先存在的全血计数(FBC),54.5%(611分中为333人)有五个可用的FBC。在我们的指导方针截止点0.30×109每晚细胞,我们在e1的嗜酸性粒细胞患者中有30.1%(581分)患病率。重复措施逐渐增加了嗜酸性患者的总比例为46.1%(581分中)E5(figure 1), with the largest increase of 7.75% (95% CI 5.71–10.23%) between E1 and E2. This trend can be also observed for other cut-off points in the literature.
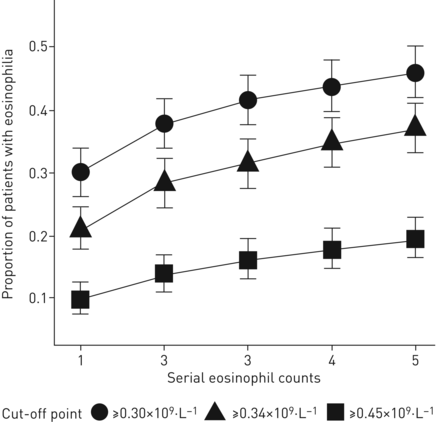
Proportion of eosinophilic patients above cut-off values with serial estimations (95% confidence intervals).
当血液嗜酸性粒细胞之间的关系d exacerbations was examined at 0.30×109每个L的细胞,E1的调节率比为1.15(0.79-1.68),p = 0.46。在E2,调节率比增加到1.67(1.16-2.39),P <0.01。进一步对E5的抽样对率没有显着的额外影响。在0.34×10观察到类似的发现9cells per L and 0.45×109cells per L cut-off points, as well as when analysis was repeated using negative binomial regression.
使用一个值来定义eosinoph血ilia is, by its very nature, arbitrary. Unsurprisingly, it is clear from our data that a greater proportion of the COPD population is defined as eosinophilic with lower cut-off points. Repeated sampling reveals an increase in the proportion of patients defined as eosinophilic in all cohorts. The rate of increase in the proportion of eosinophilic patients was dependent on the cut-off point. Thus, infigure 1, the rate of increase at 0.45×109每个L细胞小于0.30×10所示的细胞9cells per L. Further work will be needed to determine which measure provides the greatest sensitivity and specificity in reflecting the clinically important level of blood eosinophils as a biomarker. What is clear is that multiple measures are required.
Eosinophilia in E1 was not associated with an increased risk of exacerbation on multi-variable analysis. Eosinophilia in E2, E3, E4 and E5, on the other hand, had a statistically significant association with increased risk of exacerbation, even after Bonferroni correction. By taking repeated counts, the likelihood of underestimation of eosinophilia is ameliorated, but the risk of overestimation is increased. In terms of the risk of exacerbation, it appears from our analysis that two counts are required, with further estimations providing little, if any additional precision. Whether this also applies in the determination of therapeutic efficacy is unknown.
A further difficulty arises due to the different definitions of end-points. In different studies, exacerbations are variously defined and recorded, from adverse event reporting in clinical trials [2]电子记录中的编码剧集[3]。The latter are reliant on accurate data entry, coded correctly for subsequent electronic extraction. During our manual collection of data, there was a noticeable deficiency in coding community exacerbations and hospital admissions. This was particularly so in the non-numerical items that are not routinely coded. For example, we compared the accuracy of detecting hospital admissions by automated means in 50 patients. The manual search revealed eight episodes of admission in the past year; however, the automated search failed to detect a single episode.
我们的数据收集的回顾性是一个限制。不知道是否在稳定的疾病或加剧期间记录了嗜酸性粒细胞计数。然而,我们认为,我们的队列更具代表性的现实世界COPD人口,因为选择是基于临床诊断。其他潜在和回顾性队列具有严格的肌振动度标准,同时给予更加均匀的队列,可能不会反映临床医生在初级和次级护理中看到的患者。
In conclusion, we would recommend that a minimum of two or more eosinophil counts should be taken into account to determine the eosinophilic status of COPD patients. However, the eosinophil count should be considered in context, and not used as an absolute arbiter for decision making. Biomarkers, however good, are no replacements for clinical judgement.
披露
补充材料
M.G. CrooksERJ-02177-2017_CROKS.
G.A. HamadERJ-02177-2017_Hamad
A.H. Morice.ERJ-02177-2017_Morice
脚注
Support statement: This study was funded by AstraZeneca. Funding information for this article has been deposited with theCrossRef Resder注册表.
Conflict of interest: Disclosures can be found alongside this article atwww.qdcxjkg.com.
- ReceivedOctober 23, 2017.
- Accepted2017年11月6日。
- 复制right ©ERS 2018















