Abstract
Overall diet in early childhood may affect the development of respiratory symptoms. This study examined whether childhood dietary patterns are associated with respiratory symptoms in Dutch pre-school children, and whether this association could be explained by energy intake.
A prospective cohort study was performed in 2,173 children aged ≤4 yrs. Data on asthma-related symptoms were obtained by questions from the age-adapted version of the "International Study of Asthma and Allergies in Childhood" questionnaires. Data on respiratory tract infections, defined as episodes of physician attended fever with respiratory symptoms, was obtained by questionnaire. Principal components analysis was used to develop dietary patterns at 14 months of age.
Compared with low adherence, high adherence to the “Western” dietary pattern was significantly associated with frequent wheeze at 3 yrs of age (relative risk (RR) 1.39, 95% CI 1.02–1.89) and frequent shortness of breath (RR 1.44, 95% CI 1.03–2.01) and respiratory tract infections (RR 1.54, 95% CI 1.08–2.19) at 4 yrs of age. However, this association was partially explained by energy intake.
A “Western” diet may increase the risk of frequent respiratory symptoms at 3 and 4 yrs of age. In some measure, this association was explained by energy intake.
Atopic diseases are common in children and have been increasing in prevalence. Asthma is the most common chronic disease in childhood [1].Infectious diseases, particularly of the respiratory tract are a leading cause of morbidity and hospitalisation in infants and children in industrialised countries [2].Both asthma and infectious disease cause significant burden of disease in childhood [3], and identifying risk factors for the development of these diseases is of interest. The immune system undergoes substantial maturation from fetal life until childhood. The adequacy of this maturation process depends on environmental factors of which nutrition is suggested to play a role [4].Diet during pregnancy has been suggested to be associated with asthma-related symptoms in the offspring [5,6].Duration and exclusiveness of breastfeeding has been found to be related to asthma and respiratory tract infections in infancy and childhood [2,7,8].In addition, nutrition beyond the weaning period may also be of importance [4].Relatively little attention has been paid to the influence of diet beyond the weaning period on respiratory symptoms. Traditional analyses in nutritional epidemiology often examine disease in relation to specific nutrients or foods [9].Intake of specific nutrients and foods during childhood has a relationship with the development of asthma and respiratory tract infections [10,11].In addition, intake of calorie-rich foods has been associated with a higher prevalence of asthma [12].However, children eat a variety of foods with complex combinations of nutrients that are likely to be interactive or synergistic [9].Therefore, dietary pattern analysis examining the association of overall diet may be more predictive of disease risk. So far, studies examining the effect of overall diet in childhood mainly focused on a traditional Mediterranean diet. These studies found a Mediterranean diet in early life to be associated with the development of asthma-related symptoms in childhood [13,14].没有研究检查了西风的影响n diet in childhood on the development of respiratory symptoms in children. However, a Western diet has been found to be associated with an increased risk of frequent asthma attacks in adult females [15].The aim of this study was to examine whether different childhood dietary patterns are associated with respiratory symptoms in Dutch children up to 4 yrs of age. A second aim was to examine whether this association could be explained by total energy intake.
SUBJECTS AND METHODS
Participants and study design
This study was embedded in the Generation R study, a population-based prospective cohort study from fetal life until young adulthood and has been described in detail previously [16].In total, 9,778 mothers with a delivery date from April 2002 through to January 2006 enrolled in the study. Consent for post-natal follow-up was provided by 7,893 participants. Data collection on nutritional intake of the child was implemented in the study from 2003 onwards. In total, 5,088 mothers received a food-frequency questionnaire (FFQ) for their child at the age of 14 months (fig. 1). The study was approved by the medical ethical review board of the Erasmus Medical Center, Rotterdam, the Netherlands.
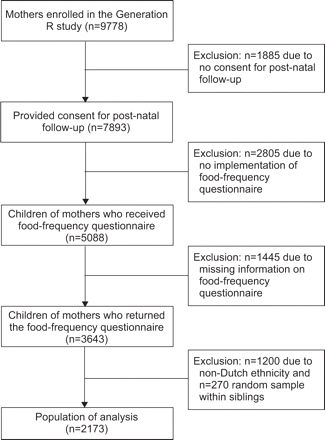
Flow chart summarising the participants of the Generation R Study.
Respiratory symptoms
Asthma-related symptoms
Data on asthma-related symptoms were obtained by questions adapted from the "International Study of Asthma and Allergies in Childhood" (ISAAC) core questionnaires on asthma at the age of 2, 3 and 4 yrs. These questions were made suitable for younger children and have been previously used in other studies [17].Asthma symptoms, including wheezing and shortness of breath, were categorised according to frequency as follows: never, 1–3 times and ≥4 times [17].Questionnaire response rates were 69%, 64% and 63% at the age of 2, 3 and 4 yrs, respectively.
Respiratory tract infections
呼吸道感染获得的数据by parent-reported questionnaires at the age of 2, 3 and 4 yrs. Respiratory tract infections were defined by the number of parent-reported physician visit(s) for fever with respiratory symptoms; coughing, runny or blocked nose, or earache. Subsequently, respiratory tract infections were categorised according to frequency as follows: never, 1–2 times and ≥3 times [17].
Dietary patterns
At the child's age of 14 months (±2 months) parents were asked to complete a FFQ. The FFQ was developed in cooperation with the division of Human Nutrition of Wageningen University, Wageningen, the Netherlands, and based on an existing validated food questionnaire described in detail previously [18], and adapted with food products frequently consumed according to a Dutch food consumption survey in infants [19].The FFQ was validated against 3-day, 24-h recalls in Dutch children at the age of 14 months. The intra-class correlation coefficients for macronutrients were as follows: total energy, 0.4; total protein, 0.7; total fat, 0.4; carbohydrates, 0.4; and dietary fibre, 0.7. The frequency of consumption of a food item was to be recorded per day, per week, or per month over the past 4 weeks. Subjects were asked to report their regular portion size relative to the standard portion size according to the Dutch table of regular portion sizes and household units [20].Total nutrient content was calculated per item according to the Dutch Nutrient Composition table [21].Response rate was 72% (n=3,643) (fig. 1).
Covariates
Variables possibly related to the outcomes such as sex, gestational age and birthweight were obtained from obstetric records assessed in midwife practices and hospital registries [16].其他潜在的混杂因素进行了评估by a combination of pre- and post-natal questionnaires completed by both parents. The questionnaires included information on maternal age, maternal socioeconomic status, maternal smoking during pregnancy, multiple parities and parental history of atopy. Maternal socioeconomic status was defined according to educational level as follows. Low: no education, primary school or <3 yrs of secondary school; mid: >3 yrs of secondary school, higher vocational training or bachelor's degree; and high: academic education. Data on breastfeeding were collected by a combination of delivery reports and post-natal questionnaires at the age of 2, 6 and 12 months. Breastfeeding was classified as (I) never, (II) exclusively for 6 months, (III) exclusively for 4 months and partially at 6 months, (IV) exclusively for 4 months, with no breastfeeding at 6 months, (V) partially for 6 months, (VI) partially for 4 months, with no breastfeeding at 6 months. Cow's milk allergy in the first year of life was assessed by questionnaire at the age of 6 and 12 months by asking parents whether their child had attended a doctor for cow's milk allergy. At the age of 12 months parents were asked about vitamin D supplementation during the past 6 months. Post-natal questionnaires completed by the mother at age 12 and 24 months included information on daycare attendance.
Population for analyses
To avoid the influence of culture differences in the definition of the dietary patterns, only children of Dutch origin were included in the analyses (n=2,443). Siblings within the Generation R cohort were randomly selected and excluded (n=270). To prevent bias associated with missing data, variables with missing values were multiple imputed (five imputations) based on the correlation between the variable with missing values with other patient characteristics [22].Consequently, n=2,173 were available after multiple imputation for statistical analyses (fig. 1).
Statistical methods
The FFQ included 211 food items which were grouped into 21 different food groups. The 21 food groups were subjected to principal components analysis (PCA). The scree plot from the PCA showed a clear break in the curve after the second component revealing the presence of two dietary patterns with Eigen values of 3.4 and 1.7. The percentage of variance explained by the dietary patterns was 16.3% and 8.2%. In the PCA, varimax rotation was used to obtain uncorrelated factors. For reasons of interpretability, the population of analysis was categorised into tertiles according to their score for the dietary pattern as follows: low, moderate and high (using “low” score as reference category).
Log-binomial regression analyses were performed with the primary outcome variables; wheezing, shortness of breath and respiratory tract infections separately at the age of 2, 3 and 4 yrs. The defined dietary patterns at the age of 14 months were analysed as primary exposure and adjusted for potential confounders. The selection of potential confounders was performed by the alteration in relative risks and kept in the multivariate model in case of an alteration of ≥10% (multivariate model 1). To assess whether the association between the dietary patterns and respiratory symptoms was explained by total energy intake additional analysis were performed with adjustment for total energy intake resulting in a separate multivariate model (multivariate model 2). The pooled results of the five imputed datasets were reported in this paper as relative risks and 95% confidence intervals. A p-value <0.05 was considered as statistically significant. The statistical analyses were performed using Stata Statistical Software for Windows, release 11 (Stata Corp., College Station, TX, USA).
RESULTS
Study population
Maternal and child characteristics of the study population are presented intable 1and the prevalence of the outcomes of interest in online supplementarytable 1.
Dietary patterns
The factor loadings of the food groups in the two dietary patterns present are shown in online supplementary table S2. The individual factor loading scores for the food groups are correlation coefficients between the food products and the specific dietary pattern. Dietary pattern 1 was associated with starchy foods, fruit, vegetables, potatoes, vegetable oils, fish, legumes and meat. This pattern is referred to here as the “Health conscious” dietary pattern. Dietary pattern 2 was associated with refined grains, soups and sauces, savoury and snacks, other fats, sugar-containing beverages and meat. This pattern will be referred to as the “Western” dietary pattern.
Dietary patterns and respiratory symptoms
High adherence to the “Western” dietary pattern was significantly associated with frequent shortness of breath (≥4) (RR 1.43, 95% CI 1.01–2.03) at age 2 yrs (table 2). High adherence to the “Western” dietary pattern was also significantly associated with frequent wheeze (≥4) (RR 1.39, 95% CI 1.02–1.89) and frequent shortness of breath (≥4) (RR 1.66, 95% CI 1.24–2.21) at age 3 yrs (tables 2and3]). However, the association between the “Western” dietary pattern and frequent shortness of breath (≥4) at the age of 2 and 3 yrs was mainly explained by maternal age, maternal socioeconomic status, maternal smoking during pregnancy, parental history of atopy, multiple parities, standard deviation score (SDS) birthweight, sex, breastfeeding, vitamin D supplementation, daycare attendance and history of cow's milk allergy (table 2). High adherence to the “Western” dietary pattern was also significantly associated with frequent wheeze (≥4) (RR 1.70, 95% CI 1.22–2.36) and shortness of breath (≥4) (RR 1.44, 95% CI 1.03–2.01) at age 4 yrs (tables 2and3]). However, the association between the “Western” dietary pattern and frequent wheeze (≥4) at age 4 yrs was mainly explained by the variables mentioned previously (table 3). Adherence to the “Western” dietary pattern was not significantly associated with frequent wheeze at age 2 yrs, or short-term wheeze (1–3 times) or shortness of breath (1–3 times) up to 4 yrs of age. High adherence to the “Western” diet was significantly associated with frequent respiratory tract infections (≥3) (RR 1.54, 95% CI 1.08–2.19) at 4 yrs of age (table 4). Adherence to the “Western” diet was not significantly associated with respiratory tract infections at 2 and 3 yrs of age or short-term respiratory tract infections (1–2 times) at 4 yrs of age. Adherence to the “Health conscious” diet was not significantly associated with respiratory symptoms up to 4 yrs of age (tables 2,3and4).
Influence of total energy intake
After adjustment for total energy intake, high adherence to the “Western” dietary pattern remained significantly associated with frequent wheeze (≥4) (aRR 1.47, 95% CI 1.04–2.07) at 3 yrs of age and frequent respiratory tract infections (≥3) (aRR 1.46, 95% CI 1.00–2.13) at 4 yrs of age (tables 3and4). After additional adjustment for total energy intake adherence to the “Western” dietary pattern was no longer significantly associated with shortness of breath up to 4 yrs of age (table 2). Adherence to the “Western” dietary pattern remained not significantly associated with any respiratory symptom at age 2 yrs or short-term respiratory symptom up to 4 yrs of age. Adherence to the “Health conscious” dietary pattern remained not significantly associated with any respiratory symptom up to 4 yrs of age (tables 2,3and4).
DISCUSSION
In this population-based prospective birth cohort study, we observed a higher risk of frequent respiratory symptoms among children at the age of 3 and 4 yrs who had a greater adherence to a “Western” diet in early childhood. However, this association was partially explained by total energy intake. No association was found between a “Health conscious” diet and respiratory symptoms up to 4 yrs of age.
Comparison with other studies of childhood dietary patterns and respiratory outcomes is difficult as most studies did not use PCA to obtain dietary patterns. However, our “Health conscious” dietary pattern (including starchy foods, fruit, vegetables, potatoes, vegetable oils, fish, legumes and meat) has some similarities with a Mediterranean diet. A study in Mexico found a Mediterranean dietary pattern to have a protective effect on asthma and asthma-related symptoms in children aged 6–7 yrs [23].A Greek study found adherence to a Mediterranean diet to be modest protective for wheezing symptoms in children aged 7–18 yrs [24].另外两名西班牙研究发现Mediterranean diet in childhood to be protective for symptoms of asthma in children aged 4 and 6–7 yrs [25,26].However, these studies were of cross-sectional design and recall bias and reverse causality might be a serious concern. To our knowledge only one prospective cohort study examined the association between a dietary pattern in childhood and atopic disease, and also found childhood adherence to a Mediterranean diet not to be significantly associated with asthma-related symptoms [14].
It has been suggested that the increase in prevalence of asthma is related to adoption of a Western lifestyle including a Western diet [27].The “Western” diet in this study was characterised by high intake of refined grains, soups and sauces, savoury and snacks, other fats, sugar-containing beverages and meat. Although no other studies examined the overall effect of a “Western-like” diet in childhood on the development of asthma-related symptoms in children, a French study did find a “Western” dietary pattern to be associated with an increased risk of frequent asthma attacks in adult females [15].Individual food components of a “Western” diet in childhood have been found to be associated with asthma symptoms in children. Sugar consumption during the perinatal period was associated with severe asthma symptoms in 6- and 7-yr-old children [28].An increased intake of saturated fatty acids was also found to be related to the risk of asthma in children [29].
Adjustment for energy intake is a standard procedure in nutritional epidemiology for standardising food and nutrient intake according to total food intake. However, most studies on dietary patterns and respiratory symptoms do not adjust for energy intake and found a Mediterranean diet to be protective for asthma-related symptoms [23,25].Chatziet al.[14] did adjust for total energy intake and found childhood adherence to a Mediterranean diet not to be significantly associated with asthma-related symptoms. An association found between a dietary pattern consisting of high energy foods and a disease outcome may not be the effect of the foods themselves, but the effect of high calorie intake. It has been suggested that high calorie foods are associated with asthma [12].Nevertheless, a Cochrane review showed only a small effect of calorie reduction on asthma [30].Indeed, after adjustment for total energy intake the “Western” dietary pattern remained significantly associated with wheeze and respiratory tract infections. Therefore, the association between the “Western” dietary pattern and respiratory symptoms was only partially explained by total energy intake and needs further elucidation.
Our large study population drawn from the general population is an important strength of this study. An additional strength is the use of dietary patterns instead of single or few nutrients or food items. The effect of single nutrients may be too small to detect as the cumulative effects of multiple nutrients in a dietary pattern may be sufficiently large. Dietary patterns identified by PCA has the advantage of reducing a large number of correlated dietary measurements down to a small number of overall dimensions of diet that are uncorrelated [9].
The time window of exposure is becoming a key aspect in the study of diseases involving systems such as the immunological and respiratory systems [13].Atopic disease often becomes manifest in early life, and it could be that the processes leading to atopic disease are initiated early in the immune development. This study assessed the effect of diet at the early age of 14 months, whereas most studies examined the effect of diet at school age. An additional strength of this study is that it examined the effect of dietary patters prior to the outcome of disease, contrary to other studies examining the effect of diet and outcome during the same time period, which may lead to recall bias and reverse causality in these studies.
一些限制必须考虑强度rpretation of our results. Information on the outcomes was obtained by parent-reported questionnaires. This could have led to misclassification of the outcome, as physician diagnosis provides more accurate outcome diagnosis. However, we do not expect this misclassification to have influenced the effect of adherence to the dietary patterns, given that the outcome was measured after the FFQ was filled out and thus it will be unlikely that the misclassification was related to the child's diet. Several arbitrary decisions are involved in identifying dietary patterns by PCA. Decisions on combining food items into food groups, the number of factors to extract, the method of rotation, and the labelling of the components may influence the reproducibility of the findings [9].Although we adjusted for potential confounders, residual confounding cannot be fully excluded, thereby precluding final conclusions regarding the causality of the study results.
Diagnosis of asthma is difficult in young children, due to the nonspecificity of the symptoms and the fact that conventional lung function tests cannot be carried out at pre-school age [17].Therefore, asthma assessment among young children is still mainly based on asthma-related symptoms. Because the diagnosis of asthma is difficult in pre-school children, our results preclude us from making conclusions regarding the presence of asthma later in life. However, Caudriet al.[17] found wheezing and serious respiratory tract infections to be predictive for the development of asthma; in particular, frequent wheezing (≥4 times per year) and frequent respiratory tract infections (≥3 times per year) were predictive of asthma at the age of 7 to 8 yrs.
In conclusion, our findings suggest that a “Western” diet may increase the risk of respiratory symptoms at the age of 3 and 4 yrs. However, this association was moderately explained by total energy intake. This study does not support a protective effect of a “Health conscious” diet on respiratory outcomes in children younger than 4 yrs of age. Further studies on respiratory outcomes to determine the association between diet and respiratory symptoms and the influence of total energy intake are worthwhile.
Acknowledgments
The Generation R Study is conducted by the Erasmus Medical Center in close collaboration with the School of Law and Faculty of Social Sciences of the Erasmus University Rotterdam, the Municipal Health Service–Rotterdam Metropolitan Area, the Rotterdam Homecare Foundation, and the Stichting Trombosedienst en Artsenlaboratorium Rijnmond. We acknowledge the contributions of children and parents, general practitioners, hospitals and midwives in Rotterdam.
Footnotes
This article has supplementary material available fromwww.www.qdcxjkg.com
Support Statement
This phase of the Generation R Study was supported by the Erasmus Medical Center, the Erasmus University Rotterdam, the Netherlands Organization for Health Research and Development (Zon Mw) and Europe Container terminals B.V. The funders had no role in the design of the study, the data collection and analyses, the interpretation of data, or the preparation of, review of, and decision to submit the manuscript.
Statement of Interest
None declared.
- ReceivedJuly 12, 2011.
- AcceptedJanuary 20, 2012.
- ©ERS 2012















