Abstract
We performed a meta-analysis to compare the impact of triple combination therapy with inhaled corticosteroids (ICS), long-acting β2-agonists (LABAs) and long-acting muscarinic receptor antagonists (LAMAs)versusLABA/LAMA combination therapy or single long-acting bronchodilator therapy in chronic obstructive pulmonary disease. The ICS/LABA/LAMA combination reduced the risk of exacerbation (relative risk 0.70, 95% CI 0.53–0.94) and improved trough forced expiratory volume in 1 s (mean difference in mL +37.94, 95% CI 18.83–53.89)versusLABA/LAMA combination therapy. The protective effect of triple combination therapyversusLABA/LAMA combination therapy against risk of exacerbation was greater in patients with blood eosinophil counts ≥300 cells·µL−1(relative risk 0.57, 95% CI 0.48–0.68). While ∼38 patients had to be treated for 1 year with ICS/LABA/LAMA combination therapy to prevent one exacerbation compared to LABA/LAMA combination therapy, the number needed to treat (NNT) was ∼21 when compared to single long-acting bronchodilator therapy. The person-based NNT per year of ICS/LABA/LAMA combination therapyversusLABA/LAMA combination therapy was significantly (p<0.05) lower in patients with eosinophil counts ≥300 cells·µL−1(NNT value: 8.58) than in those with counts <300 cells·µL−1(NNT value: 46.28). The risk of pneumonia did not differ between ICS/LABA/LAMA combination therapy and its comparators. The number needed to harm was ∼195. This meta-analysis suggests that patients on single long-acting bronchodilator therapy or LABA/LAMA combination therapy, who still have exacerbations and have blood eosinophil counts ≥300 cells·µL−1, could benefit from ICS/LABA/LAMA combination therapy.
Abstract
Triple therapy is more effective than single and dual bronchodilator therapy in reducing risk of acute exacerbation of COPD, mainly in eosinophilic patientshttp://ow.ly/9Jp630mcXJD
Introduction
Although triple combination therapy with inhaled corticosteroids (ICS), long-acting β2-agonists (LABAs) and long-acting muscarinic receptor antagonists (LAMAs) is recommended for the most severe chronic obstructive pulmonary disease (COPD) patients [1–6], a majority of the patients who receive an ICS/LABA/LAMA combination may only have mild or moderate disease [7–9]。
Since triple combination therapy should be prescribed to patients with COPD only when necessary and in concordance with treatment recommendations [9], and because there is no conclusive evidence on the superiority of triple therapy over other therapeutic options (particularly in patients at low risk of exacerbations), we performed a systematic review and a meta-analysis to determine the effect of ICS/LABA/LAMA combination therapy on the risk of exacerbation and other relevant outcomes in patients with COPD.
Methods
Search strategy
This meta-analysis has been registered in the PROSPERO database (www.crd.york.ac.uk/prospero/; registration number CRD42018095300) and performed in agreement with the Preferred Reporting Items for Systematic Reviews and Meta-Analyses Protocols (PRISMA-P) [10]。The PRISMA flow diagram is reported insupplementary figure S1. This quantitative synthesis satisfied all the recommended items reported by the PRISMA-P checklist (supplementary table S1) [10]。
Two reviewers performed a comprehensive literature search for randomised controlled trials (RCTs) evaluating the effects of ICS/LABA/LAMA combination therapy in COPD patients. The Patient problem, Intervention, Comparison, and Outcome (PICO) framework was used to develop the literature search strategy, as previously described [11]。Namely, the patient problem included “subject affected by COPD”, the intervention regarded the “administration of ICS/LABA/LAMA combination therapy”, the comparison was performed with regard to “single long-acting bronchodilator therapy and dual bronchodilator therapy” and the outcomes were the “risk of acute exacerbation of COPD (AECOPD), lung function, quality of life (QoL) and safety profile”.
The term “triple AND combination AND COPD” was searched in the Cochrane Central Register of Controlled Trials (CENTRAL), the MEDLINE, Embase, Scopus, Google Scholar, Web of Science, ClinicalTrials.gov databases and the EU Clinical Trials Register, in order to provide for relevant studies available up to May 30, 2018. No language restrictions were applied.
Citations of previously published meta-analyses and relevant reviews were examined to identify further pertinent studies, if any [12–14]。
Literature search results were uploaded to Eppi-Reviewer 4 (EPPI-Centre Software, London, UK), a web-based software programme for managing and analysing data in literature reviews that facilitates collaboration among reviewers during the study selection process.
Study selection
Published RCTs involving COPD patients that directly compared fixed or free ICS/LABA/LAMA combination therapy with fixed or free LABA/LAMA combination therapy and single long-acting bronchodilator therapy were included in this meta-analysis.
Two reviewers independently checked the relevant studies identified from literature searches obtained from the already mentioned databases. The studies were selected in agreement with the above-mentioned criteria and any difference in opinion about eligibility was resolved by consensus.
Data analysis
A network meta-analysis was performed to determine the effects of ICS/LABA/LAMA combination therapy on the primary end-points, compared with LABA/LAMA combination therapy or single long-acting bronchodilator therapy. The network meta-analysis permitted the ranking by efficacy of ICS/LABA/LAMA combination therapy, LABA/LAMA combination therapy and single long-acting bronchodilator therapy with regard to the primary end-points. A pairwise meta-analysis was also performed to confirm results obtained from the network meta-analysis and to investigate secondary end-points. Results of the pairwise meta-analysis were used to assess heterogeneity and biasviafunnel plot and Egger's test, and to quantify the relative weights of the studies included in this quantitative synthesis.
Results of the network meta-analysis are expressed as relative effect and 95% credible interval (95% CrI). Results of the pairwise meta-analysis are expressed as relative risk or mean difference and 95% confidence interval (95% CI). Since data were selected from a series of studies performed independently, a common effect size could not be assumed; thus a random-effects model was used to estimate the 95% CI for the mean distribution of drugs’ effect on the investigated variables [15]。网络分析的详细方法nd surface under the cumulative ranking curve (SUCRA) analysis are reported in thesupplementary data file.
The analysis of the number needed to treat (NNT) and number needed to harm (NNH) was performed on primary end-points. NNT and NNH are the reciprocal of the absolute risk reduction or increase, respectively, and are associated with an intervention over a fixed period of time [16–18]。The values of NNT and NNH are reported in this study as person-based and are calculated by analysing the Kaplan–Meier curves or by using the raw data provided in the primary publications, as previously described [19,20]。The relative weight of each study resulting from the pairwise meta-analysis was used to calculate the weighted average (WA) rate of the investigated arms (namely the ICS/LABA/LAMA combination arm, the LABA/LAMA combination arm and the single long-acting bronchodilator arm) and to correctly provide NNT and NNH values.
Subset analyses were focused on severe AECOPDs and blood eosinophil counts, and sensitivity analyses were performed to identify the studies that introduced heterogeneity into the findings. Meta-regression analysis was performed to examine the source of heterogeneity between-studies (I2) and to identify the factors that were associated with efficacy and safety in ICS/LABA/LAMA combination therapy with regard to primary end-points (by anchoring the efficacy and safety with comparators LABA/LAMA combination therapy or single long-acting bronchodilator therapy) [21]。The quality of the evidence was assessed for primary end-points in agreement with the Grading of Recommendations Assessment, Development, and Evaluation (GRADE) system [22]。Detailed methods used to assess the quality of RCTs and the risk of bias are reported in thesupplementary material. The OpenMetaAnalyst [23] and GeMTC [24] software was used to perform the meta-analysis, GraphPad Prism (GraphPad Software, La jolla, CA, USA) was used to graph the data and GRADEpro GDT [22] was used to assess the quality of the evidence. Statistical significance was assessed for p<0.05 and moderate to high levels of heterogeneity were considered for I2>50%.
Data extraction
Data from included RCTs were extracted and checked for study characteristics and duration, disease characteristics, age, gender, smoking habit, lung function, health-related quality of life (HRQoL), AECOPD, blood eosinophils, safety profile and Jadad score. Due to the complexity of this meta-analysis, data have been extracted in agreement with Data Extraction for Complex Meta-analysis (DECiMAL) recommendations [25]。When needed, mean and standard deviation have been estimated from the median, range and the sample size as previously described [26]。
End-points
The primary end-points of this meta-analysis were the impact of ICS/LABA/LAMA combination therapy on the risk of moderate or severe AECOPD, the change from baseline in trough forced expiratory volume in 1 s (FEV1) and the risk of pneumonia, compared to LABA/LAMA combination therapy and single long-acting bronchodilator therapy.
The secondary end-points were the impact of ICS/LABA/LAMA combination therapy on the change from baseline in St George's Respiratory Questionnaire (SGRQ) score and the risk of serious adverse events (SAEs), compared to LABA/LAMA combination therapy and single long-acting bronchodilator therapy.
Results
Study characteristics
Data obtained from 16 751 COPD patients (57.3% treated with ICS/LABA/LAMA combination therapy, 28.4% treated with LABA/LAMA combination therapy and 14.3% treated with single long-acting bronchodilator therapy) were selected from 14 studies [27–40] published between 2007 and 2018. The relevant studies and patient characteristics are described insupplementary table S2andsupplementary figure S2shows the network across the treatments involved in the Bayesian analysis.
All the RCTs subjected to meta-analysis were published as full-text papers and all but one [30] had a Jadad score of three or more [27–29,31–40]。The length of treatment ranged from 2 weeks to 52 weeks.
荟萃分析
Primary end-points
The network meta-analysis indicated that ICS/LABA/LAMA combination therapy significantly (p<0.001) reduced the risk of moderate or severe AECOPD compared to LABA/LAMA combination therapy (relative effect 0.70, 95% CrI 0.53–0.94) and single long-acting bronchodilator therapy (relative effect 0.62, 95% CrI 0.48–0.80). ICS/LABA/LAMA combination therapy also significantly (p<0.001) improved trough FEV1from baseline compared to LABA/LAMA combination therapy (relative effect 37.94 mL, 95% CrI 18.83–53.89) and single long-acting bronchodilator therapy (relative effect 68.82 mL, 95% CrI 56.95–82.48). No significant difference (p>0.05) was found for the risk of pneumonia when comparing ICS/LABA/LAMA combination therapy with LABA/LAMA combination therapy (relative effect 1.36, 95% CrI 0.84–2.00) and single long-acting bronchodilator therapy (relative effect 1.31, 95% CrI 0.76–2.32).
The SUCRA analysis indicated that ICS/LABA/LAMA combination therapy was the most effective treatment in reducing the risk of moderate or severe AECOPD and increasing trough FEV1, followed by LABA/LAMA combination therapy and single long-acting bronchodilator therapy. Conversely, the risk of pneumonia was lower with LABA/LAMA combination therapy and single long-acting bronchodilator therapy than with ICS/LABA/LAMA combination therapy. Detailed results of the SUCRA analysis and the ranking of the best therapy are reported intable 1. The WA of efficacy and safety resulting from SUCRA analysis provided the following ranking: ICS/LABA/LAMA combination therapy (WA 0.56) ≃ LABA/LAMA combination therapy (WA 0.59) >> single long-acting bronchodilator therapy (WA 0.38), where a higher WA indicates a better efficacy/safety profile.
Probability of best therapy and SUCRA values for ICS/LABA/LAMA combination therapy, LABA/LAMA combination therapy and single long-acting bronchodilator therapy in chronic obstructive pulmonary disease (COPD) patients
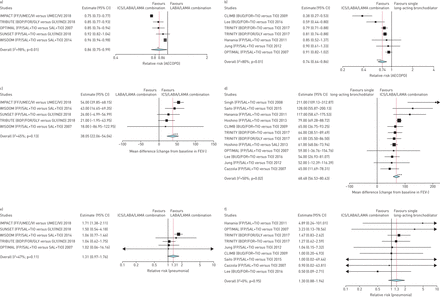
The results of the pairwise meta-analysis performed on the primary end-points overall confirmed those obtained from the network meta-analysis. Detailed results of pairwise meta-analysis are shown infigure 1.
Primary end-points: forest plots of the impact of inhaled corticosteroid (ICS)/long-acting β2-agonist (LABA)/long-acting muscarinic receptor antagonist (LAMA) combination therapyversusa, c and e) LABA/LAMA combination therapy and b, d and f) single long-acting bronchodilator therapy, a and b) on the risk of moderate or severe acute exacerbation of chronic obstructive pulmonary disease (AECOPD), c and d) on change from baseline in forced expiratory volume in 1 s (FEV1)和e和f)在慢性观察肺炎的风险tructive pulmonary disease patients. The studies have been sorted by the extent of effect. FF: fluticasone furoate; UMEC: umeclidinium bromide; VI: vilanterol; BDP: beclometasone dipropionate; FOR: formoterol fumarate; GLY: glycopyrronium bromide; IND: indacaterol; FP: fluticasone propionate; SAL: salmeterol; TIO: tiotropium bromide; BUD: budesonide. *: p<0.05versuscomparators; ***: p<0.001versuscomparators.
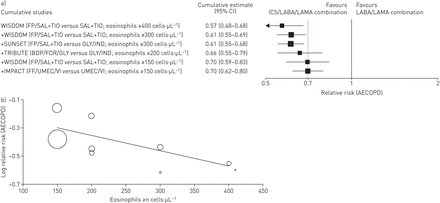
For the end-point of severe AECOPD, our analysis indicated that ICS/LABA/LAMA combination therapy was significantly (p<0.001) more effective than LABA/LAMA combination therapy (relative risk 0.78, 95% CI 0.69–0.89; I2=81%) or single long-acting bronchodilator therapy (relative risk 0.59, 95% CI 0.42–0.82; I2=82%) (supplementary figure S3). The meta-analysis showed that the protective effect of ICS/LABA/LAMA combination therapyversusLABA/LAMA combination therapy for the risk of moderate or severe AECOPD was greater in patients with higher blood eosinophil counts, ranging from a relative risk of 0.57 (95% CI 0.48–0.68) for counts of ≥400 cells·µL−1to a relative risk of 0.70 (95% CI 0.62–0.80) for counts of ≥150 cells·µL−1(figure 2a). The overall meta-regression analysis indicated that the blood eosinophil count was a significant effect modifier of ICS/LABA/LAMA combination therapy in preventing the risk of moderate or severe AECOPD (coefficient −0.001, p<0.05;figure 2b).
Cumulative studies: a) forest plot of the impact of inhaled corticosteroid (ICS)/long-acting β2-agonist (LABA)/long-acting muscarinic receptor antagonist (LAMA) combination therapyversusLABA/LAMA combination therapy on the risk of moderate or severe acute exacerbation of chronic obstructive pulmonary disease (AECOPD) in chronic obstructive pulmonary disease patients in agreement with the blood eosinophil counts, and b) meta-regression analysis of the overall impact of ICS/LABA/LAMA combination therapyversuslong-acting bronchodilators against the risk of moderate or severe AECOPD in agreement with the blood eosinophil counts. FP: fluticasone propionate; SAL: salmeterol; TIO: tiotropium bromide; GLY: glycopyrronium bromide; IND: indacaterol; BDP: beclometasone dipropionate; FOR: formoterol fumarate; FF: fluticasone furoate; UMEC: umeclidinium bromide; VI: vilanterol. *: p<0.05 for the blood eosinophil counts covariate.
The sensitivity analysis indicated that the IMPACT and WISDOM studies [27,32] represented the main source of heterogeneity with respect to the effect of ICS/LABA/LAMA combination therapyversusLABA/LAMA combination therapy for the risk of moderate or severe AECOPD. Excluding the results of these RCTs [27,32] from the meta-analysis completely abolished heterogeneity (relative risk 0.87, 95% CI 0.82–0.92; I2=0%). The sensitivity analysis performed by removing the studies of Welteet al. [36] and Leeet al. [30] reduced heterogeneity (p=0.38) when comparing ICS/LABA/LAMA combination therapyversus单一的长效支气管扩张剂治疗与注册ard to the impact on AECOPD (relative risk 0.83, 95% CI 0.78–0.88; I2=5%). Concerning the effect of ICS/LABA/LAMA combination therapyversussingle long-acting bronchodilator therapy on the change from baseline in trough FEV1, deleting the studies of Singet al. [37], Saitoet al. [31] and Hananiaet al. [35] indicated that these RCTs introduced heterogeneity in the meta-analysis (sensitivity analysis: mean difference +66.21 mL, 95% CI 58.13–74.29; I2=17%). Detailed results of the sensitivity analysis are reported insupplementary figures S4a–S4c.
Secondary end-points
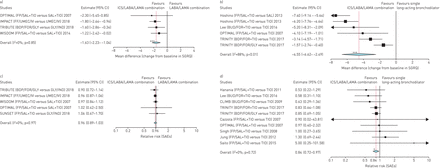
ICS/LABA/LAMA combination therapy reduced the change in SGRQ scoreversuslong-acting bronchodilators (overall effect estimate: mean difference −3.22, 95% CI −4.44 – −2.00; I2=85%), although the effect was significantly (p<0.05) greaterversussingle long-acting bronchodilator therapy (mean difference −4.20, 95% CI −6.03 – −2.37; I2=86%) thanversusLABA/LAMA combination therapy (mean difference −1.63, 95% CI −2.23 – −1.04; I2=0%) (figures 3aand 3b). The impact of triple combination therapy on the risk of SAEs is shown infigures 3cand 3d and further details are reported in thesupplementary material.
Secondary end-points: forest plots of the impact of inhaled corticosteroid (ICS)/long-acting β2-agonist (LABA)/long-acting muscarinic receptor antagonist (LAMA) combination therapyversusa和c)腊八/喇嘛联合治疗和b和d) single long-acting bronchodilator therapy, a and b) on the change from baseline in St George's Respiratory Questionnaire (SGRQ) and c and d) on the risk of serious adverse events (SAEs) in chronic obstructive pulmonary disease patients. The studies were sorted by the extent of effect. FP: fluticasone propionate; SAL: salmeterol; TIO: tiotropium bromide; FF: fluticasone furoate; UMEC: umeclidinium bromide; VI: vilanterol; BDP: beclometasone dipropionate; FOR: formoterol fumarate; GLY: glycopyrronium bromide; IND: indacaterol; BUD: budesonide. *: p<0.05versuscomparators; ***: p<0.001versuscomparators.
Number need to treat and number needed to harm analysis
The person-based NNT per year concerning the prevention of AECOPD was 38.17 for ICS/LABA/LAMA combination therapyversusLABA/LAMA combination therapy and 21.43versussingle long-acting bronchodilator therapy. The person-based NNT per year of ICS/LABA/LAMA combination therapyversusLABA/LAMA combination therapy was significantly (p<0.05) lower in patients with ≥300 cells·µL−1(NNT value: 8.58) than in those with <300 cells·µL−1(NNT value: 46.28). Further details (and 95% CI values) of person-based NNT analysis at different time points are shown intable 2and indicate that the patients that were most likely to benefit from ICS/LABA/LAMA combination therapy where those with higher blood eosinophil counts. Detailed NNT values concerning the effect of triple combination therapy on trough FEV1发表在supplementary material.
Person-based number needed to treat (NNT) at various time points concerning the prevention of acute exacerbation of COPD (AECOPD) and subset analysis based on the blood eosinophil counts in chronic obstructive pulmonary disease (COPD) patients treated with ICS/LABA/LAMA combination therapyversusLABA/LAMA combination therapy or single long-acting bronchodilator therapy
Considering pneumonia as the outcome, the person-based NNH of ICS/LABA/LAMA combination therapyversusLABA/LAMA combination therapy was 195.34 (95% CI 85.06–∞). In the subset analysis performed by considering the only RCT that included fluticasone furoate (FF) in the triple combination [27], the person-based NNH for ICS/LABA/LAMA combination therapy diminished to 33.89 (95% CI 30.69–37.84)versusLABA/LAMA combination therapy.
Meta-regression analysis
Along with blood eosinophil count, the overall meta-regression analysis indicated that Jadad score (coefficient: −0.083, p<0.05) and study duration (coefficient: −0.006, p<0.05) were significant effect modifiers of ICS/LABA/LAMA combination therapy for the analysis of moderate or severe AECOPD. No significant effect modifiers were found for trough FEV1. The meta-regression analysis indicated that the presence of FF in the ICS/LABA/LAMA combination led to significant effect modification with respect to the impact on the risk of pneumonia (coefficient: 0.46, p<0.01). In addition, FEV1values at baseline (coefficient: 0.03, p<0.05) and gender (male/female: −0.03, p<0.01) were effect modifiers for the risk of pneumonia; however, study duration and Jadad score did not significantly (p>0.05) influence the impact of ICS/LABA/LAMA combination therapy on the risk of pneumonia.
Bias and quality of evidence
Substantial levels of heterogeneity resulted for ICS/LABA/LAMA combination therapyversusLABA/LAMA combination therapy and single long-acting bronchodilator therapy with regard to the risk of moderate or severe AECOPD (p<0.001). Heterogeneity also resulted for ICS/LABA/LAMA combination therapyversusLABA/LAMA combination therapyversussingle long-acting bronchodilator therapy with respect to the change from baseline in FEV1(p<0.05).
Detailed results concerning the analysis of bias and quality of evidence are reported in thesupplementary materialassupplementary figure S5 and supplementary table S3.
Discussion
The main findings of the present meta-analysis indicated that ICS/LABA/LAMA combination therapy was significantly more effective than both LABA/LAMA combination therapy and single long-acting bronchodilator therapy in reducing the risk of moderate or severe AECOPD and improving lung function. While ∼38 patients had to be treated for 1 year with an ICS/LABA/LAMA combination to prevent one moderate or severe AECOPD compared to a LABA/LAMA combination, the NNT was ∼21 when compared to single long-acting bronchodilator therapy. The protective effect of ICS/LABA/LAMA combination therapy compared to LABA/LAMA combination therapy, relative to the risk of moderate or severe AECOPD, became greater in patients with higher blood eosinophil counts. In patients with blood eosinophil counts ≥300 cells·µL−1, the NNT for ICS/LABA/LAMA combination therapyversusLABA/LAMA combination therapy, to prevent one AECOPD in 1 year, dropped to ∼9 patients. In those with blood eosinophil counts <300 cells·µL−1, the NNT was ∼46.
No significant differences were detected with respect to the risk of pneumonia between triple combination therapy and comparators. However, the subset analysis of the RCT that included FF in the triple combination therapy [27] showed that one out of ∼34 patients treated for 1 year with a FF/umeclidinium bromide (UMEC)/vilanterol (VI) combination experienced pneumonia when compared to an UMEC/VI combination. Unexpectedly, female COPD patients seemed to be at higher risk of pneumonia and the risk of pneumonia was greater when the value of FEV1was high at enrolment.
These findings seem to support the current recommendations of the Global Initiative for Chronic Obstructive Lung Disease (GOLD) management strategy for COPD, which suggest the use of triple therapy in patients who have clinically significant symptoms and are at increased risk for frequent or severe exacerbations (GOLD group D) [6]。
Nevertheless, we must highlight that the large influence of the IMPACT study [27在荟萃分析,证明了这个团体nificant asymmetry of the funnel plot, led to a potentially biased effect estimate. The source of bias in the IMPACT study [27] is likely the enrolment of a significant percentage of patients with a history of asthma [41], such that 18% of the patients had significant bronchodilator reversibility (post-salbutamol increase in FEV1: ≥12% and ≥200 mL). However, triple therapy showed statistically significant and clinically relevant improvements on a range of important outcomes compared with LAMA/LABA combination therapy regardless of baseline reversibility [42]。
The evidence generated by this systematic review and quantitative synthesis on the available clinical evidence can help to fill, at least in part, those gaps in knowledge with regards to ICS/LABA/LAMA combination therapy and its efficacyversusLABA/LAMA combination therapy. Patients on single long-acting bronchodilator therapy or LABA/LAMA combination therapy, who still experience AECOPD and have blood eosinophil counts ≥300 cells·µL−1可能受益于三重combination therapy. Nevertheless, we must emphasise that while meta-analyses have evolved as a technique to be useful for summarising a large number of RCTs and for resolving discrepancies that are raised by those trials, they deal with populations and not with single individuals. Thus, clinicians must use clinical judgement when applying the conclusions of this and other studies to the individual patient.
Supplementary material
Supplementary Material
Please note:supplementary material is not edited by the Editorial Office, and is uploaded as it has been supplied by the author.
Supplementary materialERJ-01586-2018_Supplement
Footnotes
This article has supplementary material available fromwww.qdcxjkg.com
Author contributions: P. Rogliani provided the funding for this article. M. Cazzola and L. Calzetta completed the literature search and assessed the eligibility of studies for inclusion. All were directly involved in the acquisition of data for the article. L. Calzetta designed the statistical analyses in consultation with M. Cazzola, P. Rogliani and M.G. Matera. Statistical analyses were performed by L. Calzetta and M. Cazzola. L. Calzetta wrote the first draft of the article, in consultation with P. Rogliani and M.G. Matera for data interpretation. All authors revised the article critically for important intellectual content, gave final approval of the version to be published and agreed to be accountable for all aspects of the article in ensuring that questions related to its accuracy or integrity were appropriately investigated and resolved. L. Calzetta is the guarantor of this review and meta-analysis.
Conflict of interest: M. Cazzola has participated as a faculty member and advisor in scientific meetings and courses under the sponsorship of Almirall, AstraZeneca, Biofutura, Boehringer Ingelheim, Chiesi Farmaceutici, GlaxoSmithKline, Menarini Group, Lallemand, Mundipharma, Novartis, Pfizer, Recipharm, Verona Pharma and Zambon, and is or has been a consultant to ABC Farmaceutici, AstraZeneca, Chiesi Farmaceutici, Recipharm, Lallemand, Novartis, Ockham Biotech, Verona Pharma and Zambon. His department was funded by Almirall, Boehringer Ingelheim, Novartis and Zambon.
Conflict of interest: P. Rogliani participated as a lecturer and advisor in scientific meetings and courses under the sponsorship of Almirall, AstraZeneca, Biofutura, Boehringer Ingelheim, Chiesi Farmaceutici, GlaxoSmithKline, Menarini Group, Mundipharma and Novartis. Her department was funded by Almirall, Boehringer Ingelheim, Chiesi Farmaceutici, Novartis and Zambon.
Conflict of interest: L. Calzetta has participated as an advisor in scientific meetings under the sponsorship of Boehringer Ingelheim and Novartis, received non-financial support from AstraZeneca, a research grant partially funded by Chiesi Farmaceutici, Boehringer Ingelheim, Novartis and Almirall, and is or has been a consultant to ABC Farmaceutici, Recipharm, Zambon, Verona Pharma and Ockham Biotech. His department was funded by Almirall, Boehringer Ingelheim, Chiesi Farmaceutici, Novartis and Zambon.
Conflict of interest: M.G. Matera has participated as a lecturer and advisor in scientific meetings and courses under the sponsorship of Almirall, AstraZeneca, Boehringer Ingelheim, Chiesi Farmaceutici, GlaxoSmithKline and Novartis, and has been a consultant to ABC Farmaceutici and Chiesi Farmaceutici. Her department was funded by Novartis.
Support statement: This study was supported by institutional funds (1010107CTBOE16 University of Rome “Tor Vergata”). The funder had no role in study design, data collection, data analysis, data interpretation, or writing of the report. No sponsor had a role in this review and meta-analysis. The corresponding author had full access to all the data in the study and had final responsibility for the decision to submit for publication. Funding information for this article has been deposited with theCrossref Funder Registry.
- ReceivedJuly 5, 2018.
- AcceptedOctober 3, 2018.
- Copyright ©ERS 2018