Abstract
An epithelial–mesenchymal interaction governed by HSP60-regulated PRMT1 expression regulates airway remodelling and is reversed by bronchial thermoplastyhttp://bit.ly/2NDCKGW
Asthma is broadly defined as a group of clinical symptoms with reversible expiratory airflow limitation or bronchial hyperresponsiveness with or without airway inflammation [1].These symptoms are associated with airway remodelling, particularly in severe asthma patients, which is characterised by alterations of the tissue structure and cells within the airway, including increased submucosal extracellular matrix deposition, reticular basement membrane (RBM) thickening, airway smooth muscle (ASM) hyperplasia and hypertrophy, bronchial microvascular remodelling and mucous cell metaplasia [2–4].
Bronchial thermoplasty (BT) is an endoscopic procedure with a localised delivery of radiofrequency waves leading to heating (65°C) of the airway tissue [5].This procedure has been described as the first asthma treatment that targets airway remodelling directly instead of indirectly by modulating airway inflammation and airway hyperresponsiveness [6].Long-term follow-up studies of three randomised controlled trials pointed out that BT reduces patients' asthma exacerbations, emergency department visits and hospitalisation up to 5 years after BT treatment [7–9].This suggests that the improvement in lung function after BT might be a long-lasting effect. Indeed, BT decreased ASM mass and type I collagen deposition within the RBM [10].In addition, BT lowered submucosal nerve density, ASM-associated nerve density and the number of epithelial neuroendocrine cells. These results were associated with several clinical outcomes, including the Asthma Control Test (ACT) scores, the number of exacerbations and emergency department visits at 3 and 12 months after BT [11].The effect on the reduction in airway remodelling is likely not merely explained by a direct acute effect of thermal injury and must be due to an alternative biological mechanism triggered in response to BT [12].Although these clinical and histopathological findings have significantly expanded our knowledge on BT, and revealed that its mode of action most likely goes well beyond the originally proposed selective impact on airway smooth muscle, we still lack a detailed mechanistic understanding of how BT regulates the remodelling process.
Epithelial–mesenchymal communication is a major mechanism that regulates airway remodelling. In this issue of theEuropean Respiratory Journal, Sunet al.[13] hypothesised that crosstalk between epithelial cells and fibroblasts would underpin the beneficial effects of BT. They hypothesised that epithelial secreted factors would control epigenetic mechanisms in the fibroblast to drive long-lasting effects of BT. In their studies, they direct their attention primarily to protein arginine methyltransferase-1 (PRMT1). PRMT1 is the predominant PRMT enzyme in mammalian cells that methylates arginine residues on histones and other proteins. PRMT1 has been shown to have roles in the pathogenesis of lung cancer, pulmonary fibrosis, pulmonary hypertension, COPD and asthma [14].Deletion of PRMT1 in mouse embryonic fibroblasts leads to DNA damage and reduced cell proliferation [15].Therefore, it is of interest to study the role of PRMT1 in fibroblast proliferation leading up to airway remodelling in patients with severe asthma before and after undergoing BT.
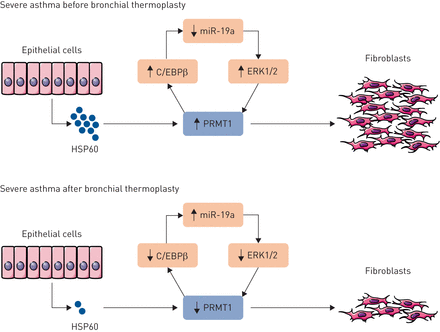
In this study, the authors collected bronchoalveolar lavage fluid (BALF) from eight patients and primary bronchial epithelial cells (PBECs) from 62 patients with severe asthma before and after undergoing BT treatment. The PBECs were grown and collected. The supernatant from PBEC culture and BALF from patients was applied to primary fibroblasts. In these airway fibroblasts, they found increased PRMT1 expression, cell proliferation and mitochondria mass in response to BALF collected before BT, which were not observed in response to BALF collected after BT. In parallel, they observed downregulation of miR-19a as well as increased extracellular signal-regulated kinase-1/2 (ERK1/2) activity in response to BALF collected before BT, whereas BALF collected after BT did not show such changes. This is of importance as miR-19a is a repressor of ERK1/2 activity and PRMT1 expression [16].CCAAT enhancer-binding蛋白质-β(C / EBPβ)你rn is a negative regulator of miR-19a [17].
The authors further report that culture supernatant from airway epithelial cells collected after BT reduced C/EBPβ expression, ERK 1/2 activity, peroxisome proliferator-activated receptor-γ coactivator-1α (PGC1α), heat shock protein-60 (HSP60), PRMT1 and mitochondria mass in airway fibroblasts compared to culture supernatant from epithelial cells obtained before BT. These results confirm that cellular crosstalk between epithelial cells and fibroblasts is key to some of the persistent changes observed after BT. Furthermore, downregulation of PRMT1 by siRNA reduced the expression of C/EBPβ and the proliferative effect of culture supernatant from epithelial cells before BT, indicating a crucial role of PRMT1 in this response. Collectively, these results show that both BALF and epithelial culture supernatant after BT decreased the proliferation of fibroblasts through a mechanism involving upregulation of miR-19a leading to downregulation of ERK1/2, PRMT1 and C/EBPβ.
To elucidate the secreted factor that initiates this communication, the authors performed proteomic analysis on BALF and transcriptomic analysis on epithelial cells. They found that HSP60 expression was downregulated after BT. HSP60 is a chaperone protein expressed in mitochondria that is involved in the transportation and refolding of proteins from the cytoplasm into the mitochondrial matrix. In addition, HSP60 is a type of protein that is induced after subjecting cells to a stress response such as heat [18].HSP60 released into the extracellular space has been described to function as a danger signal associated with autoimmune diseases, cancer, and inflammation as well as having direct interactions with molecules in various cell compartments [19].Direct stimulation of lung fibroblasts with human recombinant HSP60 significantly increased the expression of C/EBPβ, PRMT1 and PGC1α, confirming the crucial role of this intercellular signalling intermediate. Taken together, this indicates that there is an epithelial–mesenchymal interaction governed by HSP60-regulated PRMT1 expression that plays a role in airway remodelling and is reversed by BT (figure 1).
In this study, it was also found that PBECs obtained from asthma patients proliferated significantly faster after BT compared to those obtained before BT. This is in apparent contrast with previous study by Pretolaniet al. [11] who did not observe any effect of BT on regenerating epithelium. This difference is likely explained by the difference in measurement methods and timing. Pretolaniet al. [11] utilised histopathological evaluation of epitheliumin situ,whereas Sunet al.[13] used a method for quantifying proliferation of epithelial cellsex vivo. In the clinical studyin situ, the first follow-up measurement was taken after 3 months, which well exceeds the time window involved in cell proliferation of airway epithelial cells. Thus, it is not possible to infer from the Pretolaniet al. [11] study whether there were changes in epithelial cell proliferation as capturedin vitroby Sunet al.[13].
The study by Sunet al.[13] is the first one that shows the role of BT on PRMT1 expression, highlighting its role in airway remodelling. Further intriguing questions arise from this study and need to be explored. The first one is related to the mechanism of epithelial secreted HSP60 and the effects of BT hereon. It has been previously shown that stimulation by H2O2to induce oxidative stress in cells increased HSP60 expression by epithelial cells into the extracellular medium through NFκB-p65 [20].It would be of interest to study whether this is also the case with BT stimulation. It also interesting to study the epithelial secreted HSP60 signalling pathway as this appears to be a primary regulatory route towards PRMT1 expression in fibroblasts. For example, which receptors and signalling effectors are involved in fibroblast signalling in response to HSP60 and are these potential targets for drugs?
The second question is about uncovering the role of PRMT1 activity in regulating fibroblast responses. Other studies have shown that PRMT1 is responsible for modulating epithelial to mesenchymal transition through regulation of ZEB1 expression by histone modification of H4R3me2 [21] and through arginine 34 methylation of Twist1, resulting in E-cadherin repression [22].In addition, PRMT1 methylates the epidermal growth factor receptor [23].It would be of interest to also identify these possible pathways and the substrate(s) that are methylated by PRMT1 in fibroblasts, explaining its effects on airway remodelling.
Collectively, the study of Sunet al.[13] contributes to our understanding of the mechanisms involved in the beneficial effects of BT. They report sustained effects on epithelial–mesenchymal interactions in response to BT, characterised by a downregulated HSP60–PRMT1 signalling pathway. These new findings inspire not only further investigations into the pathways and epigenetic mechanisms regulated by BT, but also other potential therapeutic approaches on this signalling pathway, such as inhibition of HSP60 or PRMT1, that could potentially reduce the fibroblast responses associated with airway remodelling in asthma.
Schematic representation of the effect of bronchial thermoplasty on airway epithelial–mesenchymal communication. Bronchial thermoplasty reduces the release of heat shock protein-60 (HSP60) from epithelial cells, which in turn diminishes the expression of extracellular signal-regulated kinase-1/2 (ERK1/2), protein arginine methyltransferase-1 (PRMT1) and CCAAT enhancer-binding protein-β (C/EBPβ), whereas the expression of miR-19a is restored.
Shareable PDF
Supplementary Material
This one-page PDF can be shared freely online.
Shareable PDFERJ-01898-2019.Shareable
Footnotes
Conflict of interest: S.F. Rahmawati has nothing to disclose.
Conflict of interest: R. Gosens reports grants from Boehringer Ingelheim, Chiesi Farmaceutici and Aquilo, outside the submitted work.
Support statement: This work was supported by the Lembaga Pengelola Dana Pendidikan/Indonesia Endowment Fund for Education (LPDP). Funding information for this article has been deposited with theCrossref Funder Registry.
- ReceivedSeptember 25, 2019.
- AcceptedOctober 28, 2019.
- Copyright ©ERS 2019