抽象的
人临床Research Collaborations (CRCs) programme provides support to projects in different areas of respiratory medicine, to build and maintain pan-European and/or global collaborations. CRC applications can be submitted annually by October 15.http://ow.ly/34S730LXGNW
欧洲呼吸188bet官网地址学会(ERS)的任务是促进呼吸健康,以减轻和防止患有呼吸道疾病的疾病。为了实现这一任务,仍然需要解决许多挑战。我们需要:1)更好地了解肺部疾病及其自然史的负担,以及对疾病的基本机制;2)验证和应用准确的诊断测试,以患者为中心的结果和生物标志物;3)开发和测试新的药理和非药理干预措施及其在临床实践中的最佳实施。为了充分解决这些挑战,需要在临床医生,盟军卫生专业人员,科学家,技术和制药行业,监管机构,医疗保健提供者以及最重要的是患者之间进行合作工作。
呼吸研究财团和临床网络不是一个新概念,但通常在孤岛中工作,与ERS有可变的联系[1]. In 2013, the ERS established the Clinical Research Collaborations with the aim of: promoting the exchange of research ideas among clinicians and affiliated scientists in Europe and/or globally; building an infrastructure for prospective clinical research; securing additional funding through national and EU funding streams; and facilitating the planning, conducting, evaluation and publishing of clinical and translational studies at pan-European level and beyond.
临床,随着目前正在进行17人ch Collaborations, representing all eight major respiratory disease domains (airway diseases, interstitial lung diseases, pulmonary vascular diseases, sleep and breathing disorders, respiratory critical care, paediatric respiratory diseases, respiratory infections and thoracic oncology). These ERS Clinical Research Collaborations are linked to the respective ERS assemblies and summarised in表格1。临床研究合作申请每年可以在10月15日之前提交。每项ERS临床研究合作均可资助3年,有机会申请一个资助的续签期。ERS临床研究合作包括大量的泛欧和/或全球合作,包括针对常见和罕见肺部疾病的疾病特异性儿科和成人临床注册(后者与Ern-Lung紧密合作和合作)[2], bio-resource collections and trial platforms, as well as studies of early disease and standardisation of lung function and exercise testing. These ERS Clinical Research Collaborations map onto the eight major respiratory disease areas and the respective assemblies of the ERS as illustrated infigure 1。首届ERS临床研究合作是在支气管扩张的(Embarc [3]) and paediatric lung disease (INCIRCLE). An ERS Clinical Research Collaborations working group led by the ERS Clinical Research Collaborations Director and reporting to the Science Council assesses new applications, and oversees and supports ongoing success. Potential gaps in the ERS Clinical Research Collaborations portfolio are met by the highlight notices to encourage applications from areas of research hitherto unrepresented. Connectivity between the ERS Clinical Research Collaborations is encouraged to disseminate best practice and to align government structures. Support for the ERS Clinical Research Collaborations is provided through the research agency of the ERS Science Council [4],旨在为项目行为的协调和标准的实施做出贡献(数据隐私和数据模型,etc。). Importantly, the ERS research agency in partnership with several ERS Clinical Research Collaborations has sought external grant funding through EU funding programmes such as Horizon 2020 and the Innovative Medicines Initiative and has established successful industry collaborations.
The European Respiratory Society (ERS) Clinical Research Collaborations (CRCs)
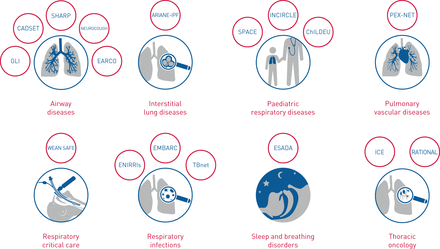
欧洲呼吸社会临床研究合作的映射188bet官网地址到八个主要呼吸道疾病区域。
人临床Research Collaborations also provide the opportunity to underpin the development and conduct of pragmatic trials. Pragmatic trials need to reflect real-life clinical practice in different healthcare settings; be generalisable and scalable to have sufficient impact on clinical practice. The ERS will endorse pragmatic trials in different respiratory disease domains that meet stringent criteria of quality and independence, and their success are likely to be predicated on either evolving from or working with the established ERS Clinical Research Collaborations [5].
欧洲肺基金会(ELF)在ERS临床研究合作中起着核心作用;ELF在工作组中是不可或缺的,并提供了支持,以确保患者参与研究,进行和交付研究。在ERS临床研究合作中强烈鼓励早期职业会员参与,以支持其职业发展,并鼓励可持续性和继任计划。
A successful ERS Clinical Research Collaboration is addressing an area of important clinical unmet need; has broad engagement to represent all stakeholders and has sufficient involvement of national respiratory societies, patients and early career members to ensure optimal implementation of the research findings into clinical practice and continuing success beyond the ERS funding period. The family of ERS Clinical Research Collaborations [6]将支持我们社会的卓越研究,并在欧洲呼吸杂志will provide details of the aims and successes of the individual Clinical Research Collaborations.
Footnotes
Conflict of interest: C. Brightling reports grant and fees for consultancy (paid to institution) from GlaxoSmithKline, AstraZeneca/Medimmune, Novartis, Boehringer Ingelheim, Chiesi and Roche/Genentech, personal fees for consultancy (paid to institution) from Vectura, Theravance, PreP, Gilead, Sanofi/Regeneron, Teva, Gossamer and 4DPharma, and institutional grants from Pfizer and Mologic, outside the submitted work.
Conflict of interest: C. Genton reports multi-sponsored study fees from Chiesi, Sanofi, Grifols, Novartis, Insmed, Zambon, TEVA and GlaxoSmithKline, outside the submitted work. C. Genton is an employee of the ERS.
利益冲突:W。Bill是ERS的执行董事。
Conflict of interest: T. Welte reports institutional grants from AstraZeneca, Bayer, Grifols, Insmed, Novartis, Pfizer and Boehringer, and personal fees for advisory board work and lectures from AstraZeneca, Bayer, Boehringer, GSK, Grifols, Insmed, Novartis, Sanofi and Pfizer, outside the submitted work.
Conflict of interest: M. Gaga has nothing to disclose.
利益冲突:E。Heuvelin报告了Chiesi,Sanofi,Grifols,Novartis,Insmed,Insmed,Zambon,Teva和Glaxosmithkline的多项赞助研究费,在提交的工作之外。E. Heuvelin是ERS的雇员。
利益冲突:G。Brusselle向顾问委员会工作报告了个人费用,并从阿斯利康,Boehringer-Ingelheim,Chiesi,Chiesi,Novartis,Glaxosmithkline和Teva发表了个人费用,以及来自Sanofi的咨询委员会工作的个人费用。
- ReceivedAugust 13, 2018.
- Accepted2018年8月18日。
- Copyright ©ERS 2018












