Abstract
COVID-19 ARDS is associated with release of biologically active neutrophil elastase-related proteinases to the airways and blood at a comparable level to non-COVID ARDShttps://bit.ly/3nihveh
To the Editor:
肺炎的严重病例经常与急性呼吸窘迫综合征(ARDS)有关,其携带约40%的死亡率[1]。Uncontrolled host inflammatory response in the lung is a key factor in the transition from pneumonia to ARDS, with alveolocapillary membrane disruption leading to interstitial and alveolar oedema [2]。Neutrophils are part of the innate immune system and are the first responders to local tissue damage and infection. Recruited neutrophils are considered important actors in lung tissue injury [3]。实际上,它们的抗菌武器的宽容武器武器可能会导致直接和间接的抵押损伤。中介粒细胞丝氨酸蛋白酶(NSP),包括弹性蛋白酶(NE),蛋白酶3(PR3)和组织蛋白酶G(CATG),并从活性细胞中释放,并在临床前和临床研究中发挥作用于ARDS病理生理学的一部分[4]。Thus, NSPs emerge as an untapped point for therapeutic interventions in pneumonia-induced ARDS [4]。These NSPs are readily synthesised in neutrophil precursors within the bone marrow and are converted into their active form by cathepsin C (CatC) [5]。They are stored together in cytoplasmic granules and secreted into the extracellular compartment upon stimulation [6]。
Coronavirus disease 2019 (COVID-19), caused by infection with severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2), can lead to pneumonia with lung hyper-inflammation [7[在最严重的情况下,对ARDS [8]。然而,有一些争议有关the clinical presentation and pathophysiology of COVID-19-induced ARDS, as compared to ARDS associated with other microbial infections. In this regard, investigation of the potential role of neutrophils in COVID-19 ARDS is only in its infancy [9–11.]。In an attempt to fill this knowledge gap, we investigated neutrophilic inflammation by studying NSPs in endotracheal aspirates (ETAs) and blood collected from mechanically ventilated patients with pneumonia-driven ARDS of non-COVID-19 and COVID-19 aetiology. CatC, identified as a biomarker of neutrophil degranulation [6使用浓缩的ETA上清液和血清中的酶和免疫方法分析促炎NSP。据我们所知,该研究是第一个比较来自不同肺炎相关ARD患者的蛋白水解活性CATC和NSP的浓度,包括Covid-19 Aetiology。
Patients from non-COVID-19 ARDS (n=15) and COVID-19 ARDS (n=17) presented similar clinical characteristics at admission. Median (interquartile range) age was 62 (57–66.5) years for the non-COVID-19 ARDS group and 64 (59–70) for the COVID-19 ARDS group, with a percentage of males of 60% and 65%, respectively. Median neutrophil count in the blood of the non-COVID-19 group was 8.7 (6.6–13.7) ×109 L-1和6.4(6.0-7.6)×109 L-1COVID-19集团。所有患者会见了criteria for ARDS, and median arterial oxygen tension/inspired oxygen fraction ratio at the time of analysis was <200 in both groups (and >100), denoting moderate severity: 197 (158–250) for the non-COVID-19 ARDS group and 162 (117–215) for the COVID-19 ARDS group. Microbial identification, using multiplex PCR and/or conventional cultures on ETA samples, was obtained in 13/15 of the non-COVID-19 patients: influenza A or B (n=8),肺炎链球菌肺炎(n = 1),鼻病毒(n = 2),呼吸合胞病毒(n = 1),葡萄球菌haemolyticus.(n = 1)和三名患有流感的流感患者被共同感染肺炎链球菌肺炎,支原体肺炎要么肺炎链球菌肺炎和aspergillus fumigatus.。
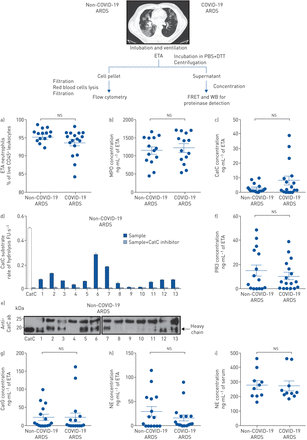
Flow cytometry analysis of the cellular content of ETAs showed that neutrophils (live CD45+/CD14-/CD16+细胞)占肺炎相关ARDS气道的绝大多数白细胞,并且注意到两组之间没有显着差异(非Covid-19 ARDS:95±0.7%; Covid-19 Ards:94±0.9%,p = 0.16)(figure 1a)。ETA neutrophils from the two groups expressed similar amount of surface activation marker CD16 (mean fluorescence intensity (MFI) for non-COVID-19 ARDS: 19930±2617; MFI for COVID-19 ARDS: 20057±2112, p=0.7340; Mann–Whitney test). Myeloperoxidase (MPO), a marker of neutrophil activation, was detected in all ETA samples of non-COVID-19 and COVID-19 ARDS patients, with mean concentrations of 1148±102 ng·mL-1和1218±111 ng·ml-1分别,两组之间的浓度没有显着差异(p = 0.63)(figure 1b)。在14名非Covid-19 ARDS患者的ETA样品中检测到CATC活性(93%; 2.6±0.8ng·ml-1)和14名Covid-19 ARDS患者(82%; 8±2.8 ng·ml-1) with no significant difference in concentration between the two groups (p=0.39) (figure 1c)。CatC activity was assayed using a CatC-selective FRET substrate [12.]在存在或不存在选择性CATC抑制剂(figure 1d)。The 20-kDa heavy chain of CatC, representing the fully processed proteolytically active subunit of the proteinase, was detected by Western blotting using an antibody against CatC in a subset of the non-COVID-19 ARDS group ETA samples. When measured activity levels were low (figure 1d), this was reflected by an absence of the heavy chain band in the corresponding blot (figure 1e)。Detection of proteolytically active CatC indicates the presence of activated neutrophils with granule release and consequently NSPs at inflammatory sites.
中性粒细胞和中性吸血物中的蛋白水解活性蛋白酶(ETAS)。工作流程的示意图如上所示。从机械通风的非Covid-19急性呼吸窘迫综合征(ARDS)和Coronavirus疾病2019(Covid-19)患者的etas称量来自重症监护单元的ARDS患者,并在PBS中孵育(5ml·g-1)在温和搅拌下,在4°C下含有1mM二硫醇(DTT)30分钟。离心后,收集上清液,然后在BCA或Bradford测定的总蛋白质定量之前浓缩五次。通过100μMCEL滤网过滤细胞粒料。使用红细胞裂解缓冲液除去红细胞,然后在染色以进行流式细胞术之前,通过40μm的细胞滤过者通过40μm的细胞滤液。用抗表面标记物(CD45,CD14和CD16)和活性染料染色细胞。中性粒细胞被定义为活CD45+CD14.-CD16+细胞。所有非Covid-19 ARDS和Covid-19 ARDS患者的ETA中的嗜中性粒细胞相对比例表示为活白细胞的百分比(CD45+cells). Individuals are depicted. The data represent mean±sem。Mann-Whitney测试。b)使用人MPO ELISA试剂盒(DMYE00B,R&D Systems)的14个非Covid-19 ARDS和14个Covid-19 ARDS患者的ETA上清液(稀释:1/500)中肌丝氧基酶(MPO)的定量检测。个人被描述。数据代表均值±sem。Mann-Whitney测试。c)通过对所有非Covid-19 ARDS和Covid-19 ARDS患者的ETA上清液和重组CATC的ETA上清液进行摩托(荧光共振能量转移)底物的水解速率,估计活性组织蛋白酶C(CATC)的浓度估计。每个点代表两个独立实验的数据,无论是重复的单一主题。个人被描述。数据代表均值±sem。Mann-Whitney测试。d)在460nm的光谱荧光测量的浓缩eta上清液(4μg蛋白)的五次浓缩η浓度的CATC活性,或没有丁腈抑制剂(L)-Thi-(L)-phe-CN(0.5μm最终,20分钟孵育37°C)使用Thi-Ala(MCA)-SER-GLY-TYR(3-NO2)-NH.2(20 µM final) as selective FRET substrate. The residual activities were inhibited by the CatC inhibitor (L)-Thi-(L)-Phe-CN which demonstrate the presence of CatC activity in samples. Inhibition experiments were performed with all ETA samples used in the study. Results with ETA samples from 13 non-COVID-19 ARDS are shown. Data represent the mean±SD.of two independent experiments, both performed with duplicates of the same sample. Similar results were obtained with ETA supernatants of all COVID-19 ARDS patients. FU: fluorescence unit. e) Western blotting (WB) of concentrated ETA supernatants (30 µg of protein) from 13 non-COVID-19 ARDS (no 1–13) using an anti-CatC antibody (sc-74590, 1:1000, Santa Cruz Biotechnology) shows the presence of the CatC heavy chain. Similar results were obtained with ETA supernatants of all non-COVID-19 ARDS and COVID-19 ARDS patients in three independent experiments. f) The proteinase 3 (PR3) activity in ETA supernatants was measured at 420 nm with or without the PR3 inhibitor Ac-PYDAP(O-C6H6-4-Cl)2(0.5 µM final, 20 min incubation at 37°C) using ABZ-VAD(nor)VADYQ-EDDnp (20 µM final) as a substrate. g) CatG activity in ETA supernatants was measured in the presence or absence of alpha-1-antichymotrypsin using ABZ-TPFSGQ-EDDnp (20 µM final) as a substrate. h) The neutrophil elastase (NE) activity in ETA supernatants was measured with or without NE inhibitor EPI-hNE-4 (0.5 µM final, 20 min incubation at 37°C) using ABZ-APEEIMRRQ-EDDnp as a substrate. The residual activities in ETA samples were inhibited by selective NSP inhibitors demonstrating the specificity of the FRET assays utilised. Inhibition experiments were performed with all ETA samples used in the study. Each point represents data of two independent experiments, both in duplicate, from an individual subject. Individuals are depicted. The data represent mean±sem。Mann-Whitney测试。i)循环NE的定量检测。人体NE ELISA套件(HK319,HYCLIZBIOTECH)用于从10个非Covid-19 ARDS和10个Covid-19 ARDS患者的血清中NE的定量检测(稀释:1/100)。个人被描述。数据代表均值±sem。Kruskal–Wallis test.ns.:无情。
The selective and specific FRET substrates designed for each NSP [13.] allowed their identification as active proteinases in ETA from patients (figure 1f–h). A reduction of NSP activities was achieved in our samples with purified alpha-1-antitrypsin, a natural serpin inhibitor of NSPs in human plasma. In this way, we were able to exclude the presence of active NSPs as a component of alpha-2-macroglobulin complexes, which encage, but still contain, the active proteinases. NSPs were detected in most samples also containing mature CatC (72%). NSP activity was detected in ETA of 12 non-COVID-19 ARDS patients (80%) and 14 COVID-19 ARDS patients (82%). We estimated the concentration of active NSPs from the rate of substrate hydrolysis using purified NSPs (non-COVID-19 ARDS group: PR3 14.8±4.4 ng·mL-1, NE 29.9±9.6 ng·mL-1, CatG 23.6±8.2 ng·mL-1; COVID-19 ARDS group: PR3 10.2±2.9 ng·mL-1, NE 16±6.2 ng·mL-1,CATG 23.7±11.7 ng·ml-1)。我们发现非Covid-19 ARDS组和Covid-19 ARDS之间的三个NSP的总浓度没有显着差异(68.4±19.7 ng·ml-1ETA和49.9±20.2 ng·mL-1ETA分别为p = 0.52)。对三个NSP的个人分析没有揭示非Covid-19和Covid-19组之间的任何差异(figure 1f–h). The variation observed in NSP concentrations could be explained by neutrophil counts in the airways, their degranulation status and the level of active extracellular inhibitors. Last, we explored circulating NSP concentration in the serum of non-COVID-19 and COVID-19 patients and compared them to healthy donors (n=10). Because of huge amounts of serpins in sera, it was not possible to detect any proteolytic activity using NSP selective FRET substrates, despite their high sensitivity. We used an ELISA (enzyme-linked immunosorbent assay) that was particularly sensitive for detection of free NE and complexed NE with alpha-1-antitrypsin. There was no significant difference in NE serum concentration between non-COVID-19 and COVID-19 groups (280±29 ng·mL-1of serum and 273±32 ng·mL-1of serum, respectively, p=1) as assayed by ELISA (n=10) (figure 1i)。However, NE concentration in both groups was significantly higher compared to healthy donors (median (interquartile range) age was 59 (54–63.5) years; male/female ratio was 2/1, n=10) (107±3.5 ng·mL-1of serum; p<0.001, Kruskal–Wallis test).
在急性炎症肺病中的蛋白水解活性NSP的特异性鉴定和检测是推断多个NSP的同时抑制作为有用且有效的治疗方法的重要起点。通过分析ETA样品,我们在非Covid-19和Covid-19患者中展示了NSP参与NSP在肺炎驱动的ARDS中的强烈的间接证据。然而,应该考虑到肺炎驱动的ARDS是肺泡过程,并且ETA采样不精确地反映肺泡舱中的展开炎症过程。尽管有这些技术限制,我们的研究表明,Covid-19 ARDS与在非Covid-19 ARDS中观察到的可比水平的生物活性NSP释放到小型气道中。据认为,NSP的水平升高了天然抑制剂的内源性盾牌。此外,我们鉴定了活性CATC,在非Covid-19和Covid-19 ARDS患者的ETA样本中,在中性粒细胞的初级颗粒中与成熟NSP一起储存的临界酶。虽然发现两组之间没有主要区别,但由于少数患者,我们不能排除低于研究的有限统计力量的差异。由于我们的观察是有限数量的患者,因为中性粒细胞可能涉及Covid-19病理生理学中的不同损害过程(包括弥漫性肺泡损伤,血栓形成和微血管病[10.]), we have not been able so far to establish a clear relationship between the outcome of pneumonia-driven ARDS patients and NSPs activities.
Taken together, our data strongly encourage further mechanistic studies with regard to the precise contribution of NSPs in pneumonia-driven ARDS and strongly support initiatives to evaluate the therapeutic potential and efficacy of CatC inhibitors in COVID-19 to eliminate NSPs already at an early phase of the disease [4]。In this regard, the outcome of an ongoing clinical trial with brensocatib, a reversible nitrile CatC inhibitor approved in a phase 2 clinical study [14.], is eagerly awaited by health professionals, scientists and currently affected patients (STOP-COVID19, Superiority Trial of Protease Inhibition in COVID-19, EudraCT number 2020-001643-13 [4]). Brensocatib treatment may help prevent irreversible pulmonary failure of COVID-19 patients with high level of active NSPs. Clinical trial results from the STOP-COVID-19 study will help us to better understand NSP-dependent pathophysiology of neutrophils in COVID-19.
Shareable PDF
致谢
The authors thank Celia Moss (Birmingham Children's Hospital and University of Birmingham) for English editing.
Footnotes
Author contributions: B. Korkmaz and S. Seren designed the study. S. Seren, L. Derian, I. Keleş, L. Gonzalez, T. Baranek and C. Paget performed the experiments. All authors analysed and discussed the results. B. Korkmaz and Y. Jouan wrote the manuscript and all authors contributed to the writing and revision process of the manuscript. B. Korkmaz supervised the work.
Conflict of interest: S. Seren has nothing to disclose.
利益冲突:L.德安没有披露。
利益冲突:I.Keleş无意义。
利益冲突:A. Guillon没有什么可披露的。
Conflict of interest: A. Lesner has nothing to disclose.
Conflict of interest: L. Gonzalez has nothing to disclose.
Conflict of interest: T. Baranek has nothing to disclose.
Conflict of interest: M. Si-Tahar has nothing to disclose.
Conflict of interest: S. Marchand-Adam has nothing to disclose.
Conflict of interest: D.E. Jenne has nothing to disclose.
Conflict of interest: C. Paget has nothing to disclose.
Conflict of interest: Y. Jouan has nothing to disclose.
利益冲突:B. Korkmaz已支付作为顾问委员会(INSMED),其他形式的咨询(新生儿治疗APS(丹麦),Santhera Pharmaceuticals(瑞士)),研讨会组织(INSMED)和在提交的工作之外旅行支持,讲座或演示。
Support Statement: This work was supported by the “Ministère de l'Enseignement Supérieur et de la Recherche”, the “Région Centre Val de Loire” (Project Pirana) and “National Science Center Poland” (UMO 2014/15/B/ST5/05311) granted to A. Lesner. Funding information for this article has been deposited with theCrossRef Resder注册表。
- 已收到October 7, 2020.
- AcceptedDecember 6, 2020.
- 版权所有©ers 2021。
This version is distributed under the terms of the Creative Commons Attribution Non-Commercial Licence 4.0.