抽象GydF4y2Ba
肺过度伸展涉及机械因素,包括大量的潮气量(GydF4y2BaVGydF4y2BaŤGydF4y2Ba),可引致发炎反应。目前的作者假设,吸气流量有助于通气诱导的炎症。GydF4y2Ba
缓冲灌注的兔肺在21%氧的情况下通气2小时GydF4y2Ba2GydF4y2Ba+ 5%COGydF4y2Ba2GydF4y2Ba,positive end-expiratory pressure of 2–3 cmH2GydF4y2BaO和随机分配:1)正常GydF4y2BaVGydF4y2BaŤGydF4y2Ba(6 mL·kg-1GydF4y2Ba)at respiratory rate (RR) 30, inspiration:expiration time ratio (I:E) 1:1, low inspiratory flow 6 mL·kg-1GydF4y2Ba·秒GydF4y2Ba-1GydF4y2Ba;2)大GydF4y2BaVGydF4y2BaŤGydF4y2Ba(12 mL·kg-1GydF4y2Ba)at RR 30, I:E 1:1, high inspiratory flow 12 mL·kg-1GydF4y2Ba·秒GydF4y2Ba-1GydF4y2Ba(HRHF);3)大GydF4y2BaVGydF4y2BaŤGydF4y2Baat RR 15, I:E 1:1, low inspiratory flow 6 mL·kg-1GydF4y2Ba·秒GydF4y2Ba-1GydF4y2Ba(LRLF);或4)大GydF4y2BaVGydF4y2BaŤGydF4y2Baat RR 15, I:E 1:2.3, high inspiratory flow 10 mL·kg-1GydF4y2Ba·秒GydF4y2Ba-1GydF4y2Ba(LRHF)。生理参数,肿瘤坏死因子(TNF)-α,白细胞介素(IL)-8和丝裂原活化蛋白激酶的活化(细胞外信号调节激酶(ERK)1/2,p38和应激活化蛋白激酶(SAPK)/的c-Jun N-末端激酶(JNK))进行了测定。GydF4y2Ba
HRHF体重增加,灌洗液IL-8和ERK1 / 2,p38和SAPK / JNK的磷酸化。这些反应是LRHF期间LRLF中缺席,但存在。在TNF-α的变化较小。组织IL-8和磷酸ERK1 / 2染色主要定位于较大细支气管和小动脉内平滑肌,外膜和支气管上皮。GydF4y2Ba
这些结果表明,在高吸气流量时,灌注肺的轻度过度伸展增强了受强湍流影响最严重的肺区域的细胞的炎症信号。GydF4y2Ba
§GydF4y2Ba这些作者对这项研究做出了同样的贡献。本文所述的研究已由美国环境保护署健康影响和环境研究实验室进行了审查,并已批准发表。批准并不意味着内容必须反映机构的观点和政策,提及商标或商业产品并不构成认可或建议使用。GydF4y2Ba
在机械通气,肺进行产生过度扩张,压缩,并在支气管和肺泡结构的剪切应力的机械力。高潮气量过度扩张(GydF4y2BaVGydF4y2BaŤGydF4y2Ba)可以产生明显的肺水肿由于物理损伤肺泡 - 毛细血管膜GydF4y2Ba1GydF4y2Ba,GydF4y2Ba2GydF4y2Ba而温和的过度扩张导致更少的直接伤害。但是,仍然可能会发生由于细胞内信号传导途径的激活和炎性细胞因子和趋化因子的随后的释放肺损伤GydF4y2Ba3GydF4y2Ba-GydF4y2Ba6GydF4y2Ba。急性肺损伤,由这种机械应力的附加损伤(呼吸机诱导性肺损伤(VILI))有助于发病率和死亡率GydF4y2Ba7GydF4y2Ba。GydF4y2Ba
VILI的机制已经在高温被归因于应力和应变由肺过度拉伸GydF4y2BaVGydF4y2BaŤGydF4y2Ba/正压通气。Ventilation of unperfused isolated rat lungs with 40 mL·kg-1GydF4y2Ba的GydF4y2BaVGydF4y2BaŤGydF4y2Ba增加趋化因子(巨噬细胞炎性蛋白(MIP)-2),在空域炎性细胞因子(肿瘤坏死因子(TNF)-α,白细胞介素(IL)-1β,IL-6)GydF4y2Ba8GydF4y2Ba。Ventilation of rats at 20 mL·kg-1GydF4y2Bafor 2 h increases MIP‐2 in the bronchoalveolar lavage (BAL) fluid9GydF4y2Ba。Ventilation of mice at 24 mL·kg-1GydF4y2Bafor 6 h increases MIP‐2 expression in lung homogenate, and in association with increased microvascular permeability and lung injury3GydF4y2Ba。Ventilation of isolated mouse lungs with 15 mL·kg-1GydF4y2Ba增加MIP-2,TNF-α和IL-6释放到灌注液GydF4y2Ba10GydF4y2Ba-GydF4y2Ba13GydF4y2Ba。GydF4y2Ba体内GydF4y2Baventilation of rabbits with 12–15 mL·kg-1GydF4y2Bafor 4 h increases TNF‐α concentration in BAL14GydF4y2Ba。Ventilation of rats at high inspiratory pressure and positive end-expiratory pressure (PEEP; 45/10 cmH2GydF4y2BaO)激活促分裂原活化蛋白激酶(MAPK)和核因子κB途径GydF4y2Ba15GydF4y2Ba。这些GydF4y2Ba体内GydF4y2Ba和GydF4y2Ba离体GydF4y2Ba效果是由许多支持GydF4y2Ba体外GydF4y2Ba研究,其中培养肺上皮细胞GydF4y2Ba6GydF4y2Ba,GydF4y2Ba16GydF4y2Ba-GydF4y2Ba19GydF4y2Ba细胞,内皮细胞GydF4y2Ba20GydF4y2Ba-GydF4y2Ba21GydF4y2Ba和巨噬细胞GydF4y2Ba五GydF4y2Ba已经经受过度拉伸。GydF4y2Ba
吸气流速也是应力在肺的重要决定因素。高吸气流量增强跨越导致动能的更大的透射到尽管相同的体积变化底层结构肺泡表面的拉伸应力。高吸气流量也剪切应力平行增大到气道和肺泡壁的表面,扭曲肺实质和变形支气管上皮细胞中的体积变化不大GydF4y2Ba22GydF4y2Ba。高吸气流量是经常需要提供足够的GydF4y2BaVGydF4y2BaŤGydF4y2Ba,但是吸气流量到肺拉伸诱导的生化反应的独立的贡献尚未确定。GydF4y2Ba
这项研究的目的是确定高吸气流速是否激活炎症细胞内信号通路。这种假设是在分离缓冲灌注肺测试通风具有中等GydF4y2BaVGydF4y2BaŤGydF4y2Ba(12 mL·kg-1GydF4y2Ba)。呼气时间比(I:E)吸气流速通过调节呼吸率(RR)和吸气变化。当前作者然后测量IL-8,MAPK的TNF-α和活化,所有这些都被链接到VILI发展的释放GydF4y2Ba3GydF4y2Ba,GydF4y2Ba8GydF4y2Ba-GydF4y2Ba15GydF4y2Ba。缓冲灌注肺系统是专门用来消除激活的非常驻炎症细胞迁移到肺产生的炎症效应。在作者的实验室中,该模型已被证明可在各种刺激下释放趋化因子/细胞因子和蛋白激酶激活GydF4y2Ba23GydF4y2Ba-GydF4y2Ba26GydF4y2Ba。GydF4y2Ba
材料与方法GydF4y2Ba
离体灌注肺标本GydF4y2Ba
该实验方案得到了杜克大学动物护理和使用委员会的批准。肺的分离和灌注是根据这个实验室发表的技术进行的GydF4y2Ba24GydF4y2Ba-GydF4y2Ba26GydF4y2Ba。新西兰大白兔(3.0-3.6 kg;罗宾逊的农场,克莱门斯,NC,美国)被肝素化和麻醉。打开胸壁,将左心房和肺动脉主干插管。主动脉也被结扎,以防止血流通过左心室。用Krebs-Henseleit‐3%白蛋白缓冲液清除循环回路中的血液。灌注回路由蓄水池、滚柱泵(Sarns, Inc., Ann Arbor, USA, MI)、气泡捕集器和与Tygon管道连接的热交换器组成。从左心房收集灌注液的蓄水池从一个力传感器(FT100型;格拉斯乐器公司,昆西,马萨,美国)。体重增加(WG)的肺是通过记录损失的液体从水库。 The fluid volume of the system was approximately 250 mL. The lungs were ventilated with 21% O2GydF4y2Ba+ 5%COGydF4y2Ba2GydF4y2Ba2-3厘米每小时GydF4y2Ba2GydF4y2Baø与动物呼吸器(哈佛仪器公司,霍利斯顿,MA,USA)。所述气体混合物含有5%的COGydF4y2Ba2GydF4y2Ba以保持CO的灌注液分压GydF4y2Ba2GydF4y2Ba的35-40 mmHg27GydF4y2Ba最大限度地减少低碳酸血症对细胞因子释放的影响GydF4y2Ba28GydF4y2Ba。再循环流量为100ml·minGydF4y2Ba-1GydF4y2Ba。采用双向Fleisch气动力计(No. 001206;连接到差压传感器(Statham, PM15, Hato Rey, PR, USA)来测量高达150ml·s的气流速率GydF4y2Ba-1GydF4y2Ba。WG,肺动脉压(GydF4y2BaPGydF4y2BaPAGydF4y2Ba),气道压力(GydF4y2BaPGydF4y2BaAWGydF4y2Ba)和空气流被记录GydF4y2Ba通过GydF4y2Ba一个4通道模拟 - 数字转换器连接到装备有数据采集软件(DATAQ仪器公司阿克伦,OH,USA)的PC计算机。最初的GydF4y2BaVGydF4y2BaŤGydF4y2Bawas 6 mL·kg-1GydF4y2Ba,产生峰GydF4y2BaPGydF4y2BaAWGydF4y2Ba的7-10 mmHg. Lungs were excluded if there were gross leaks or if thePGydF4y2BaPAGydF4y2Ba在10分钟稳定期间,>为20毫米汞柱。灌流缓冲液含NaCl (82.8 mM)、KCl (4.7 mM)、KHGydF4y2Ba2GydF4y2BaPOGydF4y2Ba4GydF4y2Ba(2.4 mM), NaHCO3GydF4y2Ba(25 mM), MgSO4GydF4y2Ba(1.2毫米),CaClGydF4y2Ba2GydF4y2Ba(2.7 mM), dextrose (11.1 mM), and bovine serum albumin (BSA), fraction V (3%, w/v).
通风协议GydF4y2Ba
肺被随机分为正常通气组和正常通气组GydF4y2BaVGydF4y2BaŤGydF4y2Ba的6 mL·kg-1GydF4y2Ba(对照组)或大GydF4y2BaVGydF4y2BaŤGydF4y2Ba的12 mL·kg-1GydF4y2Ba(table 1⇓GydF4y2Ba)。在大GydF4y2BaVGydF4y2BaŤGydF4y2Ba基,三种不同的协议被使用:1)高RR具有高吸气流量(HRHF);2)low RR with high inspiratory flow (LRHF) and 3) low RR with low inspiratory flow (LRLF) (Table 1⇓GydF4y2Ba)。这三组代表相同的高容量拉伸,但不同的吸气流量和呼吸频率。在HRHF,GydF4y2BaVGydF4y2BaŤGydF4y2Ba增加,因为RR和我这也增加吸气流速:电子持平。在LRLF,RR下降了50%以产生与对照组的下部吸气流量速率匹配。在LRHF,I:E比降低,以匹配HRHF团的高吸气流量而RR仍然很低像LRLF。Ťypical tracings of air flow and airway pressure for the four groups are shown in figure 1⇓GydF4y2Ba。空气滞留在各组缺席由零流量在呼气末(图1A-d所指示的GydF4y2Ba⇓GydF4y2Ba)。Lungs were ventilated and perfused for 120 min. Serial 2‐mL perfusate samples were taken from the effluent port and BAL was performed at the end of the experiment29GydF4y2Ba。所有流体样品储存在-80℃直至分析。GydF4y2Ba
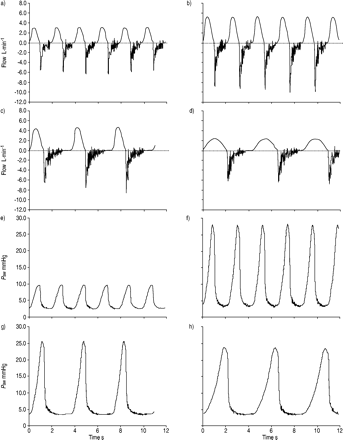
空气流(A-d)和气道压力的代表的描记(GydF4y2BaPGydF4y2BaAWGydF4y2Ba;e–h) in lungs ventilated with normal tidal volume (6 mL·kg-1GydF4y2Ba;a, e), large tidal volume (12 mL·kg-1GydF4y2Ba),分别为高流量(b, f)、低流量(c, g)、低流量(d, h)。GydF4y2Ba
通气协议GydF4y2Ba
上皮通透性的测量GydF4y2Ba
根据制造商的建议,使用牛白蛋白ELISA定量试剂盒(Bethyl Laboratories, Inc., Montgomery, TX, USA)测量牛白蛋白浓度,估算上皮通透性。该方法的最低检出限为6 ng·mLGydF4y2Ba-1GydF4y2Ba。GydF4y2Ba
肿瘤坏死因子α和白细胞介素8的测量GydF4y2Ba
浓度的肿瘤坏死因子α和IL 8在灌注液和BAL流体测量使用ELISA包根据制造商的建议(研发系统,Inc .,明尼阿波利斯,美国)。GydF4y2Ba
免疫组化GydF4y2Ba
Lungs were inflation fixed using 4% paraformaldehyde at 20 cmH2GydF4y2BaØ压力和石蜡包埋。肺切片进行脱蜡,再水化并染色IL-8和磷酸胞外信号调节激酶(ERK)1/2。初级抗体是山羊 - 抗 - 人IL-8抗体(Santa Cruz生物技术公司,Santa Cruz公司,CA,USA)和抗磷酸ERK1 / 2分别为单克隆抗体。二次抗体是生物素 - 缀合的驴抗山羊免疫球蛋白(Ig)G和山羊抗小鼠IgG分别(杰克逊ImmunoReseach Laboratories公司,西林,PA,USA)。阴性对照,用无关抗体来完成。配备有数字照相机和图像处理软件(的QCapture版本2.56;定量成像公司,伯纳比,BC,加拿大);将载玻片用Nikon显微镜(Nikon USA,梅尔维尔,NY,USA的Eclipse E600)进行了研究。GydF4y2Ba
Western blot分析GydF4y2Ba
实验结束后,在RIPA裂解缓冲液(0.1% SDS, 0.5%脱氧胆酸盐,1% Nonidet P‐40 PBS, pH 7.4)中,含抗蛋白酶鸡尾酒和1mm硫酸钒酸盐,在冰上均质化肺组织。肺匀浆离心10000×GydF4y2BaGGydF4y2Bafor 10 min, and the supernatants aliquoted. Samples were mixed with an equal volume of SDS‐PAGE loading buffer (0.125 M Tris, pH 6.8, 4% SDS, 20% glycerol, 10% β‐mercaptoethanol, and 0.05% bromophenol blue), boiled and separated on 11% SDS‐PAGE gels in Tris-glycine‐SDS buffer. Electrophoresed proteins were blotted onto nitrocellulose, blocked with 3% milk in 50 mM PBS (pH 7.4) for 1 h, washed with PBS‐0.05% Tween‐20, and incubated overnight at 4°C with primary antibodies in 5% BSA in PBS‐Tween. The primary antibodies were monoclonal antibodies against phospho‐ERK1/2, phospho‐p38, phospho‐c‐Jun N‐terminal kinase (JNK), ERK1/2, p38 and JNK/stress-activated protein kinase (SAPK) (all from Cell Signaling Technology, Inc., Beverly, MA, USA). Blots were washed and incubated with horseradish peroxidase-conjugated secondary antibody (Santa Cruz Biotechnology) in 3% milk in PBS‐Tween for 1 h at room temperature. Protein bands were detected using chemiluminescence reagents and X‐ray film (Amersham Life Science, Arlington Heights, IL, USA).
统计分析GydF4y2Ba
数据以均数±标准差表示。采用重复测量方差分析(ANOVA)对生理数据进行分析。一种方法是先用方差分析(ANOVA),然后用谢菲(Sheffe)的检验来分析数据。在进行统计的地方报告p‐值。p<0.05为差异有统计学意义。GydF4y2Ba
结果GydF4y2Ba
为了找到最优的GydF4y2BaVGydF4y2BaŤGydF4y2BaŤo stretch the lung without overtly damaging the parenchyma, two other large tidal volumes (16 mL·kg-1GydF4y2Ba和24 mL·kg-1GydF4y2Ba)were tested in preliminary experiments using RR and I:E ratio similar to those in the 12 mL·kg-1GydF4y2Ba组。Both strategies produced severe alveolar oedema within 30 min. Thus 12 mL·kg-1GydF4y2Ba被选定为大GydF4y2BaVGydF4y2BaŤGydF4y2Ba实验。GydF4y2Ba
生理效应GydF4y2Ba
Ťhe control ventilation strategy (6 mL·kg-1GydF4y2Ba)允许稳定GydF4y2BaPGydF4y2BaPAGydF4y2Ba和GydF4y2BaPGydF4y2BaAWGydF4y2Bafor 2 h with <13 g WG (table 2⇓GydF4y2Ba)。During 2 h of HRHF ventilation,PGydF4y2BaAWGydF4y2Baincreased from 8.8±0.2 mmHg to 24.3±0.9 mmHg. WG averaged 33 g or 2.5 fold that of the control group (p=0.038, interaction p<0.0001).PGydF4y2BaPAGydF4y2Baranged from 12–16 mmHg during the 2 h and was similar to that of the control group. Microvascular pressure measured by the double occlusion technique was low (data not shown). Bovine albumin concentration in the BAL fluid was not elevated (0.55±0.10 mg·mL-1GydF4y2Ba与GydF4y2Ba0.33±0.08 mg·毫升GydF4y2Ba-1GydF4y2Ba在6毫升·公斤GydF4y2Ba-1GydF4y2Ba团,p = 0.15)。GydF4y2Ba
肺动脉压(GydF4y2BaPGydF4y2BaPAGydF4y2Ba),峰值气道压力(GydF4y2BaPGydF4y2BaAWGydF4y2Ba)和weight gain in lungs ventilated with normal tidal volume (6 mL·kg-1GydF4y2Ba)、大潮气量(12ml·kg)GydF4y2Ba-1GydF4y2Ba)与高速率高流量(HRHF),低速率高流量(LRHF)和低速率低流量(LRLF)GydF4y2Ba
Lungs ventilated with LRLF had lower WG (p=0.0045, interaction p<0.0001; table 2⇑GydF4y2Ba),和略低GydF4y2BaPGydF4y2BaAWGydF4y2Ba相比于HRHF组。GydF4y2BaPGydF4y2BaPAGydF4y2Ba和BAL albumin (0.44±0.13 mg·mL-1GydF4y2Ba)均持平。该LRHF组有WG,GydF4y2BaPGydF4y2BaAWGydF4y2Ba和GydF4y2BaPGydF4y2BaPAGydF4y2Ba与HRHF相似(表2)GydF4y2Ba⇑GydF4y2Ba)。BAL albumin remained low (0.88±0.32 mg·mL-1GydF4y2Ba,P = 0.15GydF4y2Ba与GydF4y2BaHRHF;P = 0.19GydF4y2Ba与GydF4y2Ba6 mL·kg-1GydF4y2Ba)。相比于LRHF,LRLF肺有较低GydF4y2BaPGydF4y2BaAWGydF4y2Ba(table 2⇑GydF4y2Ba)。WG tended to be lower, but the difference was not statistically significant (table 2⇑GydF4y2Ba)。GydF4y2BaPGydF4y2BaPAGydF4y2Ba和BAL白蛋白也没有什么不同。GydF4y2Ba
对肿瘤坏死因子α和白细胞介素-8的效果GydF4y2Ba
HRHF通气2小时后,IL - 8的血管释放量较对照组增加约40% (p=0.035;图2GydF4y2Ba⇓GydF4y2Ba)。BAL IL-8没有差异。Compared to HRHF, lungs ventilated with LRLF had lower IL‐8 in the perfusate (p=0.044; fig. 2⇓GydF4y2Ba),类似于对照组。Lungs ventilated with LRHF had higher perfusate IL‐8 similar to the HRHF group (fig. 2⇓GydF4y2Ba)。GydF4y2Ba
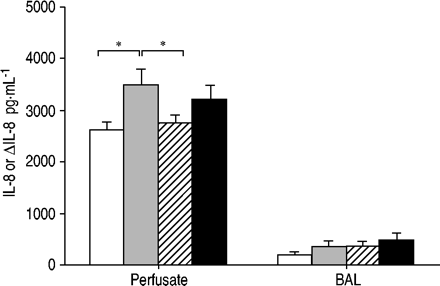
变化(Δ)白介素8 (IL)检测在灌注液和支气管肺泡灌洗液由通风与正常潮汐卷(6毫升·公斤GydF4y2Ba-1GydF4y2Ba;□), large tidal volume (12 mL·kg-1GydF4y2Ba)with high rate high flow (HRHF; ▓), low rate low flow (LRLF; └) and low rate high flow (LRHF; ▪) for 2 h. n=6 for 6 mL·kg-1GydF4y2Ba;12 mL·kg n=7GydF4y2Ba-1GydF4y2Ba+ HRHF;12 mL·kg n=6GydF4y2Ba-1GydF4y2Ba+ LRHF;和12 mL·kg n=7GydF4y2Ba-1GydF4y2Ba+ LRLF。*:P <0.05。GydF4y2Ba
灌注液中的TNF-α浓度和BAL低,并表现出四组间的最小变化。Ťhe only statistically significant change was that of BAL TNF‐α between LRLF and LRHF (fig. 3⇓GydF4y2Ba)。GydF4y2Ba
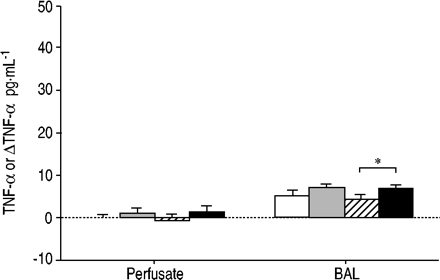
Changes (Δ) in tumour necrosis factor (TNF)‐α in the perfusate and bronchoalveolar lavage fluid produced by ventilation with normal tidal volume (6 mL·kg-1GydF4y2Ba;□), large tidal volume (12 mL·kg-1GydF4y2Ba)with high rate high flow (HRHF; ▓), low rate low flow (LRLF; └) and low rate high flow (LRHF; ▪) for 2 h. n=6 for 6 mL·kg-1GydF4y2Ba;12 mL·kg n=7GydF4y2Ba-1GydF4y2Ba+ HRHF;12 mL·kg n=6GydF4y2Ba-1GydF4y2Ba+ LRHF;和12 mL·kg n=7GydF4y2Ba-1GydF4y2Ba+ LRLF。*:P <0.05。GydF4y2Ba
HRHF resulted in strong IL‐8 staining primarily in the smooth muscle of bronchioles with an internal diameter >1 mm and associated pulmonary arterioles (fig. 4⇓GydF4y2Ba)。显著IL-8染色与LRLF通风肺缺席。GydF4y2Ba
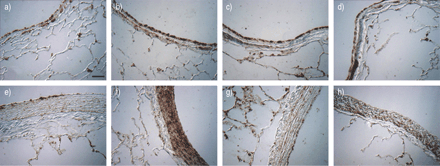
Immunocytochemistry for interleukin (IL)‐8 in the lung (a–d: bronchiole; e–h: pulmonary artery) ventilated with normal tidal volume (6 mL·kg-1GydF4y2Ba;a, e), large tidal volume (12 mL·kg-1GydF4y2Ba与高流速高流量(b, f)、低流速低流量(c, g)和低流速高流量(d, h)连用2小时。酒吧= 10µm规模。GydF4y2Ba
对丝裂原活化蛋白激酶活化作用GydF4y2Ba
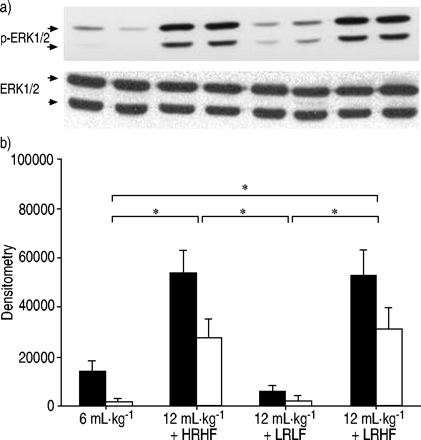
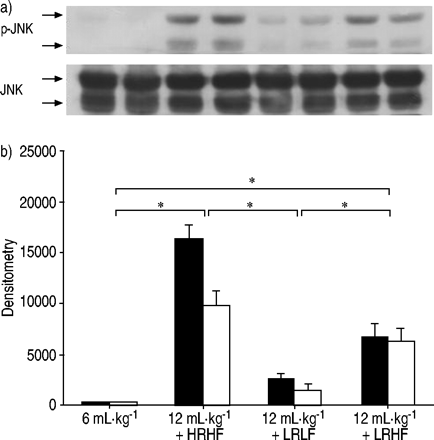
After 2 h of HRHF ventilation, MAPK activation was detected as shown by increased phosphorylation of ERK1/2 (fig. 5⇓GydF4y2Ba),p38 (fig. 6⇓GydF4y2Ba)和SAPK/JNK(图7GydF4y2Ba⇓GydF4y2Ba)。与LRHF,ERK1 / 2,p38和SAPK / JNK的磷酸化通风肺仍然增加。相反,用LRLF通风肺呈低MAPK激活类似于对照肺。GydF4y2Ba
Phosphorylation of extracellular signal-regulated kinase (ERK)1/2 after lungs were ventilated with normal tidal volume (6 mL·kg-1GydF4y2Ba)、大潮气量(12ml·kg)GydF4y2Ba-1GydF4y2Ba与高流速高流量(HRHF)、低流速高流量(LRHF)和低流速低流量(LRLF)连续工作2小时。a)磷酸化ERK1/2 (p‐ERK1/2)和总ERK1/2的代表性Western blot。每条泳道代表一个独立的实验。b) p‐ERK1/2的密度测定结果(▪:p‐ERK/1;□:p量ERK / 2)。n = 4分离实验。*:P <0.05。GydF4y2Ba
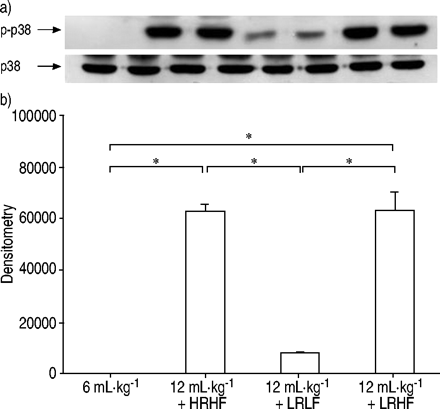
Phosphorylation of p38 after lungs were ventilated with normal tidal volume (6 mL·kg-1GydF4y2Ba)、大潮气量(12ml·kg)GydF4y2Ba-1GydF4y2Ba与高流速高流量(HRHF)、低流速高流量(LRHF)和低流速低流量(LRLF)连续工作2小时。a)磷酸化p38 (p‐p38)和总p38的代表性Western blot。每条泳道代表一个独立的实验。b) p‐p38的密度测量结果。n = 4分离实验。*:P <0.05。GydF4y2Ba
Phosphorylation of stress-activated protein kinase (SAPK)/c‐Jun N‐terminal kinase (JNK) after lungs were ventilated with normal tidal volume (6 mL·kg-1GydF4y2Ba)、大潮气量(12ml·kg)GydF4y2Ba-1GydF4y2Ba与高流速高流量(HRHF)、低流速高流量(LRHF)和低流速低流量(LRLF)连续工作2小时。A)代表性印迹磷酸化JNK(P-JNK)和总JNK。每条泳道代表一个独立的实验。b)中光密度结果的p-JNK(▪:P-JNK2 / 3;□:对JNK1)。n = 4分离实验。*:P <0.05。GydF4y2Ba
在HRHF lungs, phospho‐ERK1/2 staining increased primarily in bronchial epithelial cells and pulmonary artery smooth muscles and adventitia (fig. 8⇓GydF4y2Ba)。Similar to IL‐8, phospho‐ERK1/2 staining increased primarily in bronchioles >1 mm in internal diameter and associated pulmonary arterioles. The staining was absent in the LRLF group and intermediate in the LRHF group. There was no increased staining in alveoli or pulmonary venules (data not shown).
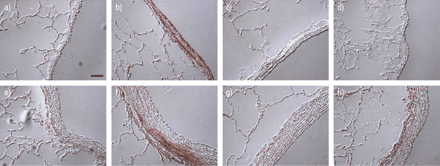
肺中磷酸化的细胞外信号调节激酶(ERK)1/2的免疫细胞化学(a-d:细支气管;e-h:肺动脉)通气,潮气量正常(6ml·kgGydF4y2Ba-1GydF4y2Ba;a, e), large tidal volume (12 mL·kg-1GydF4y2Ba与高流速高流量(b, f)、低流速低流量(c, g)和低流速高流量(d, h)连用2小时。酒吧= 10µm规模。GydF4y2Ba
讨论GydF4y2Ba
这项研究的主要的新发现是HRHF通气量增加肺IL-8的释放和激活肺MAPK,两者LRLF通气期间缺席。虽然在IL-8和MIP-2的变化已经在分离的肺的其他研究中注意到GydF4y2Ba10GydF4y2Ba,GydF4y2Ba12GydF4y2Ba,GydF4y2Ba三十GydF4y2Ba-GydF4y2Ba33GydF4y2Ba和肺的通气与中等大GydF4y2BaVGydF4y2BaŤGydF4y2Ba(up to 20 mL·kg-1GydF4y2Ba)GydF4y2Ba体内GydF4y2Ba9GydF4y2Ba,GydF4y2Ba34GydF4y2Ba中,不受控的吸气流速。IL-8是非常重要的,因为它是一个强有力的中性粒细胞趋化因子是在脓毒症患者和持续性急性呼吸窘迫综合征增加GydF4y2Ba35GydF4y2Ba-GydF4y2Ba37GydF4y2Ba。此外,增加的抗IL-8水平的:IL-8复合物与死亡率增加有关GydF4y2Ba38GydF4y2Ba。IL-8是也血管生成和诱导增殖和内皮细胞和平滑肌细胞的趋化性GydF4y2Ba39GydF4y2Ba。在肺中的几个驻留细胞可以产生IL-8与机械应力,包括上皮细胞GydF4y2Ba五GydF4y2Ba,GydF4y2Ba6GydF4y2Ba,GydF4y2Ba18GydF4y2Ba与肺泡巨噬细胞GydF4y2Ba五GydF4y2Ba。在目前的研究中的免疫化学染色表明,大的肺小动脉和细支气管的平滑肌细胞也IL-8释放的来源。GydF4y2Ba
越来越多的证据体表明MAPK激活是早期事件在从mechanosensors信令细胞核以刺激基因转录GydF4y2Ba40GydF4y2Ba。ERK、p38和SAPK/JNK的快速磷酸化已被观察到在循环拉伸的肺细胞中GydF4y2Ba16GydF4y2Ba,GydF4y2Ba18GydF4y2Ba,GydF4y2Ba41GydF4y2Ba。Phosphorylation of ERK1/2 and SAPK/JNK was found in adult rats ventilated with an injurious high pressure (peak inspiratory pressure/PEEP=45/10 cmH2GydF4y2BaO)GydF4y2Ba15GydF4y2BaERK1/2和SAPK/JNK在肺泡上皮细胞中被激活。在目前的研究中,肺通气时吸气峰压/PEEP (35/3 cmH)较低GydF4y2Ba2GydF4y2BaO)还激活了ERK1/2、p38和SAPK/JNK。ERK1/2的活化在较大的肺小动脉和细支气管中最为显著。细胞分布的差异可能与肺拉伸损伤的程度相对于血流剪切应力的大小有关(见下文)。GydF4y2Ba
也许本研究最重要的发现是在HRHF IL-8释放和MAPK激活可通过减少吸气流量(LRLF)来防止。在充气期间,肺实质和呼吸道受到toboth拉伸应力和剪切应力GydF4y2Ba42GydF4y2Ba。拉伸应力垂直作用于表面,其大小是由跨肺压来确定。剪切应力作用在主要平行的方向上的表面上,剪切应力与层流的大小由粘度,平均流速和所述气道的直径确定:GydF4y2Ba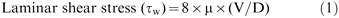 其中μ是空气的粘度;V是平均流速(体积流量除以管区域,πDGydF4y2Ba2GydF4y2Ba/ 4);和d是管的直径。当前研究的HRHF协议中使用的GydF4y2BaVGydF4y2BaŤGydF4y2Ba的12 mL·kg-1GydF4y2BaŤo ventilate the lung at 30 breaths per min and an I:E ratio of 1:1 resulting in an average inspiratory flow of 12 mL·kg·s-1GydF4y2Ba。假设空气的粘度为1.73×10GydF4y2Ba-5GydF4y2BaN-S·米GydF4y2Ba-2GydF4y2Ba,Ťhe laminar shear stress at the rabbit trachea (internal diameter=6 mm) during HRHF ventilation would be ∼0.03 N·m-2GydF4y2Ba。在大气道其中流动主要是湍流的,雷诺应力占主导地位,并且可以是若干百倍大于切应力(3-10 N·m的高GydF4y2Ba-2GydF4y2Ba)。Even in lower airways with an internal diameter of 0.6 mm (8–10th generation) where the volumetric flow rate has fallen significantly, wall shear stress may still be significant, especially at airway bifurcations and curvature43GydF4y2Ba。Ťhis concept is supported by the increased IL‐8 and ERK1/2 immunostaining in bronchioles >1 mm in internal diameter. The current findings did not exclude the contribution of lung oedema or the higher velocity of perpendicular stress applied to alveolar surface in the HRHF group to IL‐8 release and the degree of MAPK activation. However, this effect would be much less than in previous studies where very large tidal volumes were used and tissue destruction was pronounced10GydF4y2Ba,GydF4y2Ba12GydF4y2Ba,GydF4y2Ba三十GydF4y2Ba-GydF4y2Ba33GydF4y2Ba。GydF4y2Ba
其中μ是空气的粘度;V是平均流速(体积流量除以管区域,πDGydF4y2Ba2GydF4y2Ba/ 4);和d是管的直径。当前研究的HRHF协议中使用的GydF4y2BaVGydF4y2BaŤGydF4y2Ba的12 mL·kg-1GydF4y2BaŤo ventilate the lung at 30 breaths per min and an I:E ratio of 1:1 resulting in an average inspiratory flow of 12 mL·kg·s-1GydF4y2Ba。假设空气的粘度为1.73×10GydF4y2Ba-5GydF4y2BaN-S·米GydF4y2Ba-2GydF4y2Ba,Ťhe laminar shear stress at the rabbit trachea (internal diameter=6 mm) during HRHF ventilation would be ∼0.03 N·m-2GydF4y2Ba。在大气道其中流动主要是湍流的,雷诺应力占主导地位,并且可以是若干百倍大于切应力(3-10 N·m的高GydF4y2Ba-2GydF4y2Ba)。Even in lower airways with an internal diameter of 0.6 mm (8–10th generation) where the volumetric flow rate has fallen significantly, wall shear stress may still be significant, especially at airway bifurcations and curvature43GydF4y2Ba。Ťhis concept is supported by the increased IL‐8 and ERK1/2 immunostaining in bronchioles >1 mm in internal diameter. The current findings did not exclude the contribution of lung oedema or the higher velocity of perpendicular stress applied to alveolar surface in the HRHF group to IL‐8 release and the degree of MAPK activation. However, this effect would be much less than in previous studies where very large tidal volumes were used and tissue destruction was pronounced10GydF4y2Ba,GydF4y2Ba12GydF4y2Ba,GydF4y2Ba三十GydF4y2Ba-GydF4y2Ba33GydF4y2Ba。GydF4y2Ba
流体引起的剪切应力在0.5〜5 N·m的范围内GydF4y2Ba-2GydF4y2Ba激活MAPK,增强炎性细胞因子的释放,诱导基因表达,并调节在血管内皮细胞骨架对准和细胞增殖和平滑肌细胞GydF4y2Ba20GydF4y2Ba,GydF4y2Ba44GydF4y2Ba-GydF4y2Ba51GydF4y2Ba。然而鲜为人知的是,关于由高气流对肺细胞或在肺中产生的剪切应力的生化效应。高吸气流量应该产生在较大的气道上皮细胞周围结构上的上皮的影响最大和高切向力产生相对于内部结构质膜的水平偏转,GydF4y2Ba如。GydF4y2Ba细胞骨架成分GydF4y2Ba52GydF4y2Ba。这种失真可以激活变形敏感的膜 - 和细胞骨架 - 介导的机械传导GydF4y2Ba53GydF4y2Ba。采用有限元分析,赖福和KallokGydF4y2Ba54GydF4y2Ba所研究的动脉和支气管之间的相互作用,并发现扭曲力施加到两个结构围绕支气管或动脉产生非均匀的应力。的应力集中在孔隙空间邻接动脉和支气管的平均动脉周围和支气管周的径向应力数次。这些分析表明在支气管壁的应力可以被发送到邻近的动脉GydF4y2Ba通过GydF4y2Ba间质和该bronchoarterial护套内的间质可以是泪肺过度充气期间的焦点。的丰富的IL-8和MAPK在目前的研究中高流动染色和周围的肺动脉(平滑肌和外膜)提供了证据这一假说。负责是不知道的mechanosensors;对支气管上皮细胞,这样的结构不明确的,但候选物包括整联蛋白受体GydF4y2Ba55GydF4y2Ba,阿米洛利敏感性钠通道GydF4y2Ba56GydF4y2Ba-GydF4y2Ba58GydF4y2Ba,也许还有纤毛GydF4y2Ba52GydF4y2Ba。GydF4y2Ba
HRHF对IL-8和MAPK的突出影响是在对比对TNF-α的影响相对较小。这可能是HRHF通气时的机械应力可能没有足够的触发TNF-α释放。使用较大的几项研究GydF4y2BaVGydF4y2BaŤGydF4y2Ba在BAL证明增加TNF-αGydF4y2Ba8GydF4y2Ba,GydF4y2Ba31GydF4y2Ba,GydF4y2Ba33GydF4y2Ba而其他人没有GydF4y2Ba三十GydF4y2Ba,GydF4y2Ba59GydF4y2Ba,GydF4y2Ba60GydF4y2Ba。这是可能的TNF-α释放通过同时损伤加剧,GydF4y2Ba如。GydF4y2Ba,内毒素GydF4y2Ba17GydF4y2Ba,GydF4y2Ba三十GydF4y2Ba或缺血GydF4y2Ba8GydF4y2Ba。在隔离肺缺乏一个完整的支气管循环也可能影响细胞反应。GydF4y2Ba
总之,目前的作者报告说,吸气流量是轻度肺病的拉伸过程中血管白细胞介素-8释放和促分裂原活化蛋白激酶激活的重要机械决定因素,而对细胞发生较大的支气管肺结构内的主要效果。在受伤肺部受到机械通气,在拉伸应力大幅增加是由增加的表面张力引起的重复打开和关闭ofunstable肺泡单元GydF4y2Ba42GydF4y2Ba。的呼气末压力的应用程序通过防止不稳定的肺泡去复张减轻一些这些应力GydF4y2Ba34GydF4y2Ba。研究表明,高吸气流速也可刺激较大的气道和肺小动脉释放炎性介质,并且减少吸气流速可能会提供额外的保护肺。GydF4y2Ba
致谢GydF4y2Ba
作者感谢美国杜克大学医学中心和J.绍库普的L.塔特罗,美国环境保护署的J.卡特的技术支持。这项研究由NHLBI(P01-HL073997)wassupported通过赠款。GydF4y2Ba
- 收到GydF4y2Ba二零零三年十一月一十九日。GydF4y2Ba
- 公认GydF4y2Ba2004年3月10日。GydF4y2Ba
- ©ERS期刊有限公司GydF4y2Ba
参考GydF4y2Ba
- ↵GydF4y2Ba
- ↵GydF4y2Ba
- ↵GydF4y2Ba
- ↵GydF4y2Ba
- ↵GydF4y2Ba
- ↵GydF4y2Ba
- ↵GydF4y2Ba
- ↵GydF4y2Ba
- ↵GydF4y2Ba
- ↵GydF4y2Ba
- ↵GydF4y2Ba
- ↵GydF4y2Ba
- ↵GydF4y2Ba
- ↵GydF4y2Ba
- ↵GydF4y2Ba
- ↵GydF4y2Ba
- ↵GydF4y2Ba
- ↵GydF4y2Ba
- ↵GydF4y2Ba
- ↵GydF4y2Ba
- ↵GydF4y2Ba
- ↵GydF4y2Ba
- ↵GydF4y2Ba
- ↵GydF4y2Ba
- ↵GydF4y2Ba
- ↵GydF4y2Ba
- ↵GydF4y2Ba
- ↵GydF4y2Ba
- ↵GydF4y2Ba
- ↵GydF4y2Ba
- ↵GydF4y2Ba
- ↵GydF4y2Ba
- ↵GydF4y2Ba
- ↵GydF4y2Ba
- ↵GydF4y2Ba
- ↵GydF4y2Ba
- ↵GydF4y2Ba
- ↵GydF4y2Ba
- ↵GydF4y2Ba
- ↵GydF4y2Ba
- ↵GydF4y2Ba
- ↵GydF4y2Ba
- ↵GydF4y2Ba
- ↵GydF4y2Ba
- ↵GydF4y2Ba
- ↵GydF4y2Ba
- ↵GydF4y2Ba
- ↵GydF4y2Ba