To the Editor:
We read with pleasure the article “Serum CC chemokine ligand-18 predicts lung disease worsening in systemic sclerosis” [1),酒吧lished in theEuropean Respiratory Journal. Tievet al. [1] demonstrated very nicely that elevated CC chemokine ligand 18 (CCL18) serum levels predict lung disease progression in patients with systemic sclerosis (SSc). In the multivariate analysis, the hazard ratio for lung function worsening or death was 5.36 for SSc patients with serum CCL18 concentration above 187 ng·mL−1. They provided clear evidence of this in a French cohort of 83 SSc patients, yet a second affirmation cohort was missing. Recently, Elhajet al.[2] showed a rather weak correlation of CCL18 levels with changes in forced vital capacity (FVC) over time studying a large cohort of early SSc patients, but failed to analyse lung function data in regards to progression of SSc-associated interstitial lung disease (ILD) accordingly to European Respiratory Society (ERS) consensus. Moreover, neither cut-off values for CCL18 levels nor hazard ratios were reported by the authors. Thus, the results of Tievet al. [1] still need to be validated in a second, independent cohort.
Systemic sclerosis (SSc) is a multi-organ connective tissue disease, in which heart and lung involvements are the most common causes of death. Published 10-year survival rates vary between 55% and 77% [3].New therapies as well as new biomarkers, reflecting disease activity and prognosis, are therefore urgently needed.
Fibrotic lung diseases are characterised by a distinct macrophage activation type called alternative or M2 [4].M2 macrophages are key players in wound healing, fibrosing processes and are similar to tumour-associated macrophages (TAMs) [5].CCL18 is mainly secreted by human myeloid cells, especially by macrophages [6] and is one of the most important human markers of alternatively activated macrophages. Elevated CCL18 levels are described in various fibrotic diseases of the lung,e.g. systemic sclerosis and idiopathic interstitial pneumonias [7], especially idiopathic pulmonary fibrosis [8].
To determine the predictive relevance of serum CCL18, we correlated CCL18 with pulmonary function tests and survival data in a German cohort of 96 patients with SSc. SSc diagnosis was established according to American College of Rheumatology criteria [9].All included SSc patients attended the outpatient clinic of the tertiary referral centre at the University hospital Charité Berlin, Germany, and were followed-up for 2 years. The study was approved by the local ethics committee. During routine check-up (at least once a year), pulmonary function tests were performed, according to the American Thoracic Society (ATS) recommendations using a body plethysmograph. A decline of more than 10% predicted in forced vital capacity compared with baseline was considered a significant decline, corresponding to the ATS/ERS criteria [10].To quantify CCL18 serum concentrations, we used the DuoSet ELISA Development System Kit (R&D Systems Europe, UK) pursuant to manufacturer's protocol. CCL18 cut-off values were established using receiver operating characteristic curves comparing the areas under the curve. We computed Kaplan–Meier curves and performed (univariate and multivariate) Cox proportional hazards models to analyse the influence of CCL18 on time to lung function worsening, defined as decline of the predicted forced vital capacity (FVC), 10% or death.
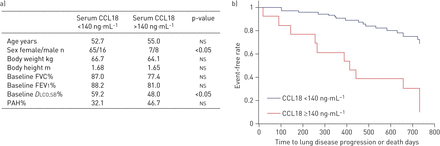
Our results were as follows. Basic patient's characteristics are displayed infigure 1a. On average, baseline serum CCL18 concentration was 101±62 ng·mL−1and ranged from 27 ng·mL−1to 421 ng·mL−1. 15 patients died during follow up. The optimal cut-off for baseline serum CCL18 concentration was 140 ng·mL−1, based on the maximal area under the curve in the receiver operating characteristic analysis. 15 SSc patients had a serum concentration above 140 ng·mL−1. SSc patients with serum CCL18 concentrations above 140 ng·mL−1did not significantly differ from SSc patients with concentrations lower than 140 ng·mL−1in age, body weight and height, baseline FEV1or in baseline FVC, but significantly in sex and single-breath diffusing capacity of the lung for carbon monoxide. SSc patients with CCL18 levels above 140 ng·mL−1had a significantly higher risk for death or lung function worsening (p<0.0001) within 2 years than SSc patients with low CCL18 levels. The Kaplan–Meier curve is depicted infigure 1b. The hazard ratio to suffer from death or progression of SSc-associated ILD was 4.8 in the univariate Cox hazard model and 5.1 in the multivariate Cox hazard model (after adjusting for age, sex, baseline FVC, body weight and height), respectively.
a) Basic patient characteristics. Data are presented as mean, unless otherwise stated. FVC: forced vital capacity; FEV1: forced expiratory volume in 1 s;DLCO,SB:次呼吸的肺的扩散能力carbon monoxide; PAH: pulmonary arterial hypertension; ns: not significant. b) Kaplan–Meier Survival analysis of SSc patients (n = 96) grouped by serum CCL18 concentrations. The blue line represents the group of SSc patients with serum CCL18 under 140 ng·mL−1 and the red line represents the group of SSc patients with over 140 ng·mL−1, respectively.
In conclusion, we were able to confirm in a second independent cohort, that serum CCL18 concentration is a significant predictor of mortality and progression of ILD in patients with SSc. This holds true even after adjusting for physiological parameter. The optimal CCL18 cut-off value in our cohort was 140 ng·mL−1 and hence lower compared to the 187 ng·mL−1 determined by Tiev et al, maybe due to technical differences in CCL18 measurement. However, the hazard ratios of the univariate (HR=4.8 vs. 5.4) and the multivariate (HR=5.1 vs. 5.4) analysis were very similar. This demonstrates that, in scleroderma, CCL18 has the potential to become a major biomarker for clinical evaluation, therapy planning and stratification in clinical trials.
Acknowledgments
The authors would like to thank N. Wehrle and S. Kamenker (Dept of Pneumology, Medical Centre, University Hospital Freiburg, Germany) for skilful technical assistance.
Footnotes
Conflict of interest: None declared.
- ReceivedJuly 30, 2013.
- AcceptedSeptember 17, 2013.
- ©ERS 2014